Rank the following substituted phenols in order of increasing acidity, and explain your answer: CH
Question:
Rank the following substituted phenols in order of increasing acidity, and explain your answer:
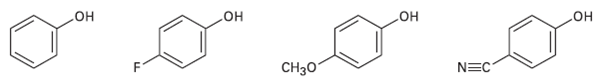
Transcribed Image Text:
CHзо но но но он
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
Remember that electronwithdrawing groups stabilize phenox...View the full answer

Answered By

BETHUEL RUTTO
Hi! I am a Journalism and Mass Communication graduate; I have written many academic essays, including argumentative essays, research papers, and literary analysis. I have also proofread and written reviews, summaries and analyses on already finished works. I am eager to continue writing!
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Rank the following complex ions in order of increasing wavelength of light absorbed. Co(H20+ CoCN)-, Col, Colen)
-
Rank the following in order of increasing acidity for the indicated hydrogen atoms (bold) (1 = least acidic; 4 = most acidic). (a) (b) (c) O C OCH3 . CH3 H3C
-
Rank the following five compounds in order of increasing acid strength: 2-chloroethanol, p-chlorophenol, p-methylphenol, ethanol, and phenol.
-
Which of the following is not an element of the fraud triangle? (a) Rationalization. (b) Financial pressure. (c) Segregation of duties. (d) Opportunity.
-
How does environmental analysis at the domestic level differ from global analysis?
-
The accountant for a subunit of Speed Sports Company went on vacation before completing the subunits monthly performance report. This is as far as she got: Requirements 1. Complete the performance...
-
How long will it take money to quadruple (i.e., increase by 300 percent) in an account earning an 8 percent EAR? (You may assume that interest is paid smoothly and continuously throughout the year,...
-
Dansyl chloride, which absorbs maximally at 330 nm and fluoresces maximally at 510 nm, can be used to label amino acids in fluorescence microscopy and FRET studies. Tabulated below is the variation...
-
If you are able to earn 4 percent interest, what amount would you need to invest to have $1,950 two years from now? Use time value of money factors with at least four decimal places and then round...
-
A block with mass M = 5.0kg rests on a frictionless table and is attached by a horizontal spring (k = 130 N/m) to a wall. A second block, of mass m = 1.25 kg, rests on top of M. The coefficient of...
-
Benzoquinone is an excellent dienophile in the DielsAlder reaction. What product would you expect from reaction of Benzoquinone with 1 equivalent of 1, 3-butadiene from reaction with 2 equivalents of...
-
Benzyl chloride can be converted into benzaldehyde by treatment with nitro methane and base. The reaction involves initial conversion of nitro methane into its anion, followed by SN2 reaction of the...
-
Silas is about to begin regular month-end contributions of $500 to a bond fund. The funds long-term rate of return is expected to be 6% compounded semiannually. Rounded to the next higher month, how...
-
Assume that you are the leadership team of a convenience store chain that has more than 300 outlets. The company is facing an inventory shrinkage problem, and store managers report that the main...
-
Gamers Technology ( GT ) is a private company with a December 3 1 st fiscal year end. GT is an online magazine which it sells on a 1 2 - month subscription basis. To meet the daily demands of its...
-
You, CPA, work for Mercer Consulting Ltd. It is now February 12, 2019, and your boss, Patricia Mercer, has just returned from a meeting with Adam Burke of Burke Entertainment Ltd. (BEL). Patricia...
-
function addChar ( input , character ) { if ( input . value = = null | | input.value = = " 0 " ) input.value = character else input.value + = character } function cos ( form ) { form.display.value =...
-
Simplify. 7x x 4 x+2 2 x+3 x-9x + 14 x+3x
-
What is the difference between robots and robotics?
-
TRUE OR FALSE: 1. Banks with a significantly large share of fixed-interest rate home loans are less exposed to interest rate risks. 2. Although Australian banks are pretty big, they are not...
-
Show that the formation of NaCl 2 (s) is very unfavorable; that is, fH[NaCl 2 (s)] is a large positive quantity. Assume that the lattice energy for NaCl 2 would be about the same as that of MgCl 2 ,...
-
Classify the following solvents as being protic or aprotic: formic acid, HCO2H; acetone, CH3COCH3; acetonitrile, CH3CN; formamide, HCONH2; sulfur dioxide, SO2; ammonia, NH3; trimethylamine, N(CH3)3;...
-
Would you expect the reaction of propyl bromide with sodium cyanide (NaCN), that is, CH3CH2CH2Br + NaCN CH3CH2CH2CN + NaBr to occur faster in DMF or in ethanol? Explain your answer.
-
When tert-butyl bromide undergoes solvolysis in a mixture of methanol and water, the rate of solvolysis (measured by the rate at which bromide ions form in the mixture) increases when the percentage...
-
Which of the following statements accurately describe risk? Select all that apply. 1 point If compromised, a medium-risk asset may cause some damage to an organization's finances. Organizations often...
-
The following question comes from Chapter 2 of the course textbook, Economics and Financial Management for Nurses and Nurse Leaders (3rd ed., 2017), by S. J. Penner and are edited for formatting. An...
-
Margot reviews the financial statements of a potential investment target. She notices that in the company's multiple-step income statement a separately listed gain is reported as part of "income from...

Study smarter with the SolutionInn App


