A sample consisting of 1 mol of perfect gas atoms (for which CVm = 3/2 R) is
Question:
A sample consisting of 1 mol of perfect gas atoms (for which CV•m = 3/2 R) is taken through the cycle shown in Fig. 2.34.
(a) Determine the temperature at the points 1, 2, and 3.
(b) Calculate q, w, ∆U, and ∆H for each step and for the overall cycle. If a numerical answer cannot be obtained from the information given, then write in +, -, 0, or? As appropriate
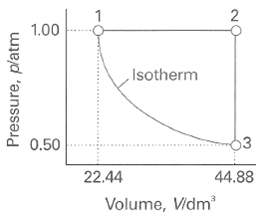
Transcribed Image Text:
1.00 ,Isotherm 13 0.50 22.44 44.88 Volume, Vidm' Pressure, platm
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
The temperatures are readily obtained from the perfect gas equation T PV nR 100 atm x 224 dm 100 mol ...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physical Chemistry questions
-
Gas within a chamber passes through the cycle shown in Figure. Determine the energy transferred by the system as heat during process CA it the energy added as heat QAB during process AB is 20.0 J, no...
-
A monatomic ideal gas is taken around the cycle shown in Fig. 20.29 in the direction shown in the figure. The path for process c → a is a straight line in the pV-diagram. (a) Calculate Q, W, and...
-
A sample consisting of 2.00 mol of perfect gas molecules, for which CV, m= 5/2 R, initially at PI = 111 kPa and TI = 277 K, is heated reversibly to 356 K at constant volume. Calculate the final...
-
M1 is a way to measure... a) the level of bank reserves b) a country's money supply c) the level of savings in a country d) a country's economic potential
-
Discuss how the Frank-Starling law of the heart helps to explain the influence of venous return on stroke volume.
-
Draw a line-bond structure for propane, CH3CH2CH3. Predict the value of each bond angle, and indicate the overall shape of the molecule.
-
Who can sign a pleading?
-
Sally Fleming operates her own catering service. Summary financial data for February are presented in equation form as follows. Each line designated by a number indicates the effect of a transaction...
-
Cottage Company deposits all cash receipts on the day they are received, and it makes all cash payments by check. At the close of business on December 31, its Cash account shows a debit balance of...
-
A maker of energy drinks is considering abandoning can containers and going exclusively to bottles because the sales manager believes customers prefer drinking from bottles. However, the vice...
-
Given that = 1.11 K atm-I for carbon dioxide, calculate the value of its isothermal Joule- Thomson coefficient. Calculate the energy that must be supplied as heat to maintain constant temperature...
-
A sample consisting of2.0 mol CO2 occupies a fixed volume of 15.0 dm3 at 300 K. When it is supplied with 2.35 kJ of energy as heat its temperature increases to 341 K. Assume that CO2, is described by...
-
Valley Corporation is attempting to select the best of a group of independent projects competing for the firms fixed capital budget of $4.5 million. The firm recognizes that any unused portion of...
-
3. A circular parallel plate capacitor of radius R = 1 cm is connected to a time-dependent voltage, V(t) = (10V) sin (wt), with a frequency of f = 100 Hz. The capacitance of the capacitor is constant...
-
Below is the complete list of accounts of of a company and the related balance at the end of April. All accounts have their vormal debit or credit balance, Cash, $3,500, Prepaid Rent, $7,000;...
-
The firm believes that its stock price at December 31, 20X11, does not accurately reflect its intrinsic value on the same date. Assume that 20X11 dividends were $31,049. Calculate the intrinsic value...
-
Using the direct stiffness method, determine the reactions at the supports. Draw shear & moment diagrams. Members are axially rigid. While recovering the member internal forces, first use slope...
-
A 10 MVA, 22 kV star connected alternator has armature resistance of 0.5 ohm. The machine is supplying 90 A current at zero pf leading when the terminal voltage is 25 kV. Under this condition, if the...
-
In the Fig. 28 shown crank \(A B\) is \(15 \mathrm{~cm}\) long and is rotating at \(10 \mathrm{rad} / \mathrm{s} . C\) is vertically above \(A\). \(C A\) equals \(24 \mathrm{~cm} . C\) is swivel...
-
Write a declaration for each of the following: a. A line that extends from point (60, 100) to point (30, 90) b. A rectangle that is 20 pixels wide, 100 pixels high, and has its upper-left corner at...
-
Understand how planning occurs in todays organizations.
-
Refer to the information in Exercise 6.15(b) and sketch the cooling curves for liquid mixtures in which x(B 2 H 6 ) is (a) 0.10, (b) 0.30, (c) 0.50, (d) 0.80, and (e) 0.95. Data in Exercise 6.15(b)...
-
Methane (melting point 91 K) and tetrafluoromethane (melting point 89 K) do not form solid solutions with each other, and as liquids they are only partially miscible. The upper critical temperature...
-
Show that two phases are in mechanical equilibrium only if their pressures are equal.
-
Given that 8g(x)+7x sin(g(x)) = 8x + 41x - 42 and g() = 0, find g(). d' (33) =
-
In order to develop the model of a production system that contains a milling machine, we gathered the processing times of 1,000 parts that got processed from it. These data values were used directly...
-
Purpose of Conducting Financial Analysis Financial analysis is the process of evaluating businesses, projects, budgets, and other finance-related transactions to determine their performance and...

Study smarter with the SolutionInn App


