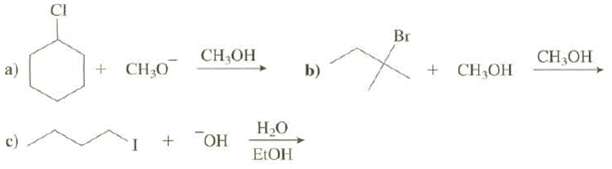
Show both the substitution and elimination products that are formed in these reactions: a) C CI +
Question:
Show both the substitution and elimination products that are formed in these reactions:
Transcribed Image Text:
a) C CI + CH₂0 CH₂OH + OH H₂O EtOH b) Br + CH₂OH CH3OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
Elimination reactions compete with substitution reaction...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show both the substitution and elimination products that would be formed in these reactions: to , -CI a) Br CH,OH CH,O b) CI E:OH + CH;CH,OH
-
Give the substitution and elimination products you would expect from the following reactions. (a) 3-bromo-3-ethylpentane heated in methanol (b) 1-iodo-1-methylcyclopentane heated in ethanol (c)...
-
What substitution and elimination products (if any)might be obtained when each of the following alkyl halides is treated with sodium methoxide in methanol? (a) methyl iodide (b)...
-
Describe, in human terms, why delay and jitter are bad in real time (interactive) voice and video communications. Would these same problems apply to recorded voice and video stored and played back at...
-
The Launceston Wool Company designs and manufactures woollen coats, which are in high demand the cold Tasmanian winters. The company uses a process costing system to cost products because 1: produces...
-
First answer the following two questions about your preferences: a. You are given $ 5,000 and offered a choice between receiving an extra $ 2,500 with certainty or flipping a coin and getting $ 5,000...
-
The transactions below are for the business Consulting Services Ltd owned by Jere Duckin. The business began on 1 September 20x8 and has been operating for one month. The following transactions took...
-
Profit Corporation authorized Anderson, an employee, to find a buyer for used equipment that Profit intended to sell. Anderson believed that he had authority to contract for the sale of the...
-
A group of 2418 students were surveyed about the courses they were taking at their college with the following results: 1034 students said they were taking History. 1281 students said they were taking...
-
Allie has bought a new apple orchard. The orchard has a single file of trees, numbered from 1 to N. Each tree has a certail number of ripe apples. Allie has a rule she wants to follow. She wants to...
-
Show the products of these reactions. (Remember that acid-base reactions are usually much faster than nucleophilic substitution reactions.) a) C1-CHCHCHCOH b) Br OH NH3 + OH
-
Show the rearranged carbocations that are expected from these carbocations: a) +CH CH3 b) CHCHCHCHCH c) CH3
-
Figure 9-23 shows a cylindrical riser attached to a casting. Compare the solidification times for each casting section and the riser and determine whether the riser will be effective?
-
Ten years ago, Mobiletex Ltd. Leased a patent for the manufacture of a gadget used in mobile phones from Quickcom Ltd. The lease agreement provided for payment of royalty at the rate of Sh.70 per...
-
Define Management Accounting and the components of the Manufacturing Statement.
-
A woman invests $6400 in an account that pays 6% interest per year, compounded continuously. (a) What is the amount after 2 years? (Round your answer to the nearest cent.) $ 7215.98 (b) How long will...
-
How do you pass a file name as an argument in python? What is the built in input function in python? Assist me in writing a small function named punctuate that takes an input parameter named txt ,...
-
Question 2 [8] Suppose you take a short position in a three-month futures contract on 2 500 kilograms of Fine Wool at a futures price of 1 010 cents per kilogram. You are required to deposit R19 000...
-
When pension funds and insurance funds invest money, where does the money come from?
-
A fuel pump sends gasoline from a car's fuel tank to the engine at a rate of 5.88 10-2 kg/s. The density of the gasoline is 735 kg/m3, and the radius of the fuel line is 3.18 10-3 m. What is the...
-
Evaluate each using the values given. (-(-5-x)); use x = 6, and = = -2
-
Reaction of acetone with D 3 O + yields hexadeuterioacetone. That is, all the hydrogen?s in acetone are exchanged for deuterium. Review the mechanism of mercuric ion?catalyzed alkyne hydration, arid...
-
Give IUPAC names for the following alkylhalides: (b) (a) CHCH2CH2CH21 H C (c) C,2I BrCH2CH2CH2CH2Br CH (e) Br (1) 1 -CH2CI CI CH (d) CH2H2CI CHH2H CHCH2CH2CH I
-
Draw structures corresponding to the following IUPAC names: (a) 2-Chloro-3, 3-dimethylhexane (b) 3, 3-Dichloro-2-methylhexane (c) 3-Bromo-3-ethylpentane (d) 1, 1-Dibromo-4-isopropylcyclohexane (e)...
-
Which of the following are correct ways of recording partnership equity? Method 1 Method 2 i Partner's Drawings account is closed off to: Patner's capital account Partner's retained earnings...
-
Because absorption costing capitalises fixed manufacturing overhead costs to inventory, managers using it may build up inventories unnecessarily. True False
-
. How financial assets and financial liabilities are recorded? Write your answer in 200-300 words. Explain the following cost management tools and techniques in 200-250 words. a) Cost Estimation b)...

Study smarter with the SolutionInn App


