For the system described in Problem 11.47, and using the tube size found in that problem, compute
Question:
Transcribed Image Text:
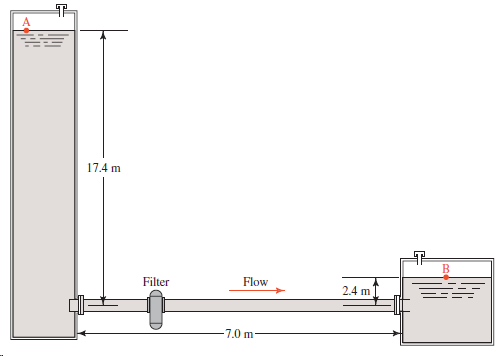
17.4 m Flow Filter 2.4 m -7.0 m
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
APPLIED FLUID MECHANICS Objective Volume flow rate Problem 1148 Figure 1133 Propyl alcohol at 25C Sy...View the full answer

Answered By

Joseph Njoroge
I am a professional tutor with more than six years of experience. I have helped thousands of students to achieve their academic goals. My primary objectives as a tutor is to ensure that students do not have problems while tackling their academic problems.
4.90+
10+ Reviews
27+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Suppose that for the system described in Problem 129, the spring constants are each k = 60 N/m. The system starts from rest and slowly accelerates until the masses are 0.8 m from the center of the...
-
For the pumping system described in Problem 14.23, how will the maximum elevation above the surface of the reservoir change if the water temperature is 80C(P v = 47.35 kPa)? Data From Problem 14.23 A...
-
For the steam power plant described in Problem 11.1, assume the isentropic efficiencies of the turbine and pump are 85% and 80%, respectively. Find the component specific work and heat transfers and...
-
Let (x) = x 2 + 3x + 2 and g(x) = x + 1. Find each of the following. (a) (fg)(x) (b) (fg) (-2) (c) (+)(x) (d) ()(-2)
-
Justine argues that all companies should use the revaluation model as it provides more useful information than the cost model. She is also concerned that, if companies do have a choice, they will...
-
Selected transactions for Tara Lawn Care Company are listed below. 1. Made cash investment to start business. 2. Paid monthly rent. 3. Purchased equipment on account. 4. Billed customers for services...
-
For a test of H0: 1 = 2 versus H1: 1 2, the sample sizes were n1 = 15 and n2 = 25. How many degrees of freedom are there for the test statistic? Use the simple method.
-
Following are several transactions that relate to the Village of Brooks Haven in 2012. Prepare all the journal entries necessary to record these transactions. In addition, identify the fund( s) in...
-
The PostHoc Company specializes in justifying decisions that have already been made. One day they fielded a request to perform a project risk analysis and compute the risk factor for buying an...
-
On June 1, John Sullivan opened a real estate office in Hamilton called Sullivan Realty. The following transactions were completed for the month of June. Note that facsimile documents have been...
-
For the system described in Problem 11.47, and using the tube size found in that problem, compute the expected volume flow rate through the tube if the pressure above the fluid in the large tank at A...
-
Figure 11.33 shows a part of a chemical processing system in which propyl alcohol at 25°C is taken from the bottom of a large tank and transferred by gravity to another part of the system. The...
-
A 240-m 3 rigid tank is filled with a saturated liquidvapor mixture of water at 200 kPa. If 25 percent of the mass is liquid and 75 percent of the mass is vapor, the total mass in the tank is (a) 240...
-
Discuss the importance of duty to warn for behavioral health professionals.
-
Describe how spirituality and religion help to shape ones healthcare choices.
-
Describe the objectives of tort law.
-
Describe common legal issues for behavioral health professionals.
-
What forms of negligence are described in this chapter?
-
In the interleaving approach to FEC, assume each packet contains 10 samples from a sampled piece of music. Instead of loading the first packet with the first 10 samples, the second packet with the...
-
In 1995 Miguel purchased a home for $130,000. In 2000 he sold it for $170,000 and immediately purchased another one for $180,000, which he sold in 2007 for $235,000. How much taxable capital gain, if...
-
When the following compound is treated with sodium ethoxide, two condensation products are obtained, both of which are produced via Dieckmann cyclizations. Draw both products. OEt Eto
-
For each of the following reactions, predict the major product and propose a mechanism for its formation. (a) (b) (c) :? 1) LDA 2) CH3I 1) NaH 2) -CH,Br
-
Identify the reagents you would use to achieve the following transformation:
-
The froghopper Philaenus spumarius is supposedly the best jumper in the animal kingdom. To start a jump, this insect can accelerate at 4.00 km/s over a distance of 2.0 mm as it straightens its...
-
find T(625). I Given the recurrence relation T(n)=7T (n/5)+ 10n for n > 1 T (1)=1 Answer: (please write your answer here, add required space if needed)
-
Given a string length of 1.00 0.01 m and a period of 2.00 0.10, what is the uncertainty in gravity? No need to include units, but give them as m/s^2.

Study smarter with the SolutionInn App


