For the tank shown in Fig. 3.21, determine the reading of the bottom pressure gage in psig
Question:
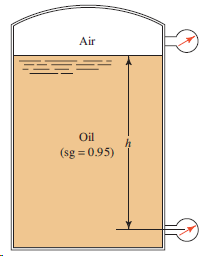
Transcribed Image Text:
Air Oil (sg = 0.95)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 82% (17 reviews)
p108 psig yh108 p...View the full answer

Answered By

Gaurav Soni
Teaching was always an area where I can pursue my passion. I used to teach my friends and junior during my school and college life. After completing my professional qualification (chartered accountancy) and before joining my job, I also joined an organization for teaching and guidance to my juniors. I had also written some articles during my internship which later got published. apart from that, I have also given some presentations on certain amendments/complex issues in various forms.
Linkedin profile link:
https://www.linkedin.com/in/gaurav-soni-38067110a
5.00+
7+ Reviews
13+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For the tank shown in Fig. 3.21, determine the depth of the oil h if the reading of the bottom pressure gage is 35.5 psig, the top of the tank is sealed, and the top gage reads 30.0 psig. Air Oil (sg...
-
For the tank shown in Fig. 6.38, compute the velocity of flow from the outlet nozzle at varying depths from 10.0 ft to 2.0 ft in 2.0-ft increments. Then, use increments of 0.5 ft to zero. Plot the...
-
For the tank shown in Fig. 6.25, calculate the volume flow rate of water from the nozzle. The tank is sealed with a pressure of 20 psig above the water. The depth h is 8 ft. Air under pressure Water...
-
1. You are required to compare the results of your printed reports and the results of reports of the MYOB assignment book in your MYOB assignment. If it is assumed that two results of the reports are...
-
Rotterdam University sold 10,000 season football tickets at 90 each for its five-game home schedule. What entries should be made, (a) When the tickets were sold, and (b) After each game?
-
Consider the element silicon, Si. (a) Write its electron configuration. (b) How many valence electrons does a silicon atom have? (c) Which subshells hold the valence electrons?
-
TekCare Corp. is considering purchasing one of two new diagnostic machines. Either machine would make it possible for the company to bid on jobs that it currently isn't equipped to do. Estimates...
-
If the ending inventory of a firm is overstated by $50,000, by how much and in what direction (overstated or understated) will the firms operating income be misstated?
-
You are overseeing a corporate restructuring that involves closing a manufacturing plant in rural Ohio (note that no remote work opportunities are available - must report to work in-person). The...
-
Creeping flow around a spherical bubble, when a liquid flows around a gas bubble, circulation takes place within the bubble. This circulation lowers the interfacial shear stress, and, to a first...
-
For the tank shown in Fig. 3.21, determine the reading of the bottom pressure gage in psig if the top of the tank is sealed, the top gage reads 50.0 psig, and the depth of the oil h is 28.50 ft. Air...
-
For the tank in Fig. 3.22, compute the depth of the oil if the depth of the water is 2.80 m and the gage at the bottom of the tank reads 52.3 kPa(gage). Vent Oil (sg = 0.86) h2 Water
-
The machine shop at Bycraft Enterprises operates 24 hours a day and uses a numerically controlled (NC) welding machine. The load on the machine is monitored, and no more than 24 hours of work is...
-
Write an essay e Eastman Kodak Company downfall. The essay must be written in the form of an analysis for the CEO/board and recommend economic responses. It is important that the essay uses, and...
-
A scale is fixed to the end of a shaft of torsional stiffness 2.5 N-m/rad. A viscous damping torque of magnitude 1.5 Nm resists the motion of the pointer on a scale at an angular velocity of 2 rad/s....
-
A new hotel company (the entrant) is planning on building a new hotel next year adjacent to an incumbent hotel company which, to date, has had a monopoly on the hotel market, earning $100 million in...
-
As indicated in your materials, many view the common-law approach to determining employment as problematic. For one thing, the resulting tests are highly fact intensive and therefore often create...
-
On June 1, 2022, John purchased a new X-ALU Sportscar from Sam's Dealership which specializes in selling vehicles manufactured by ALU Motors at their auto plant in Glendale, California. On June 13,...
-
Express each statement as an equation involving the indicated variables. The volume V of a cube is the cube of the length x of a side.
-
In a system with light damping (c < cc), the period of vibration is commonly defined as the time interval d = 2/d corresponding to two successive points where the displacement-time curve touches one...
-
Dogs cool off in hot weather by panting. Write a chemical equation to describe this process and calculate H o R.
-
Use the following data at 298.15 K to complete this problem: Calculate ÎH o R for a. OH(g) H(g) + O(g) b. H 2 O(g) 2H(g) + O(g) c. H 2 O(g) H(g) + OH(g) Assuming ideal gas behavior, calculate...
-
Identify the alkyne you would use to prepare each of the following compounds via hydroboration-oxidation: (a) (b) (c)
-
Harrison Forklift's pension expense includes a service cost of $27 million. Harrison began the year with a pension liability of $47 million (underfunded pension plan). Interest cost, $8; expected...
-
Jackson Company adquiere el 100 % de las acciones de Clark Corporation el 1 de enero de 2020 por $4100 en efectivo. A esa fecha, Clark tiene el siguiente balance de prueba: Dbito Crdito Efectivo $...
-
What is not a reason for a company to buy back its common stock in the open market? 1.It is trying to get rid of hostile shareholders. 2.It believes that the stock is underpriced. 3.There are future...

Study smarter with the SolutionInn App


