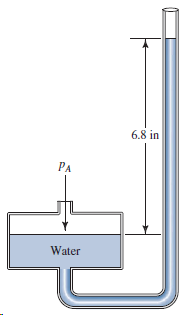
For the well-type manometer in Fig. 3.34, calculate p A . 6,8 in PA Water
Question:
Transcribed Image Text:
6,8 in PA Water
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
Patm 68 in ...View the full answer

Answered By

Muhammad Ahtsham Shabbir
I am a professional freelance writer with more than 7 years’ experience in academic writing. I have a Bachelor`s Degree in Commerce and Master's Degree in Computer Science. I can provide my services in various subjects.
I have professional excellent skills in Microsoft ® Office packages such as Microsoft ® Word, Microsoft ® Excel, and Microsoft ® PowerPoint. Moreover, I have excellent research skills and outstanding analytical and critical thinking skills; a combination that I apply in every paper I handle.
I am conversant with the various citation styles, among them; APA, MLA, Chicago, Havard, and AMA. I also strive to deliver the best to my clients and in a timely manner.My work is always 100% original. I honestly understand the concern of plagiarism and its consequences. As such, I ensure that I check the assignment for any plagiarism before submission.
4.80+
392+ Reviews
587+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Figure 3.35 shows an inclined well-type manometer in which the distance L indicates the movement of the gage fluid level as the pressure pA is applied above the well. The gage fluid has a specific...
-
The inclined manometer in Fig P2.37 contains Merriam red oil, SG = 0.827. Assume the reservoir is very large. If the inclined arm has graduations 1 inch apart, what should θ be if each...
-
For the compound differential manometer in Fig. 3.32, calculate (p A - p B ). Water, Oil (sg = 0.90) 6 in 8 in 6 in 10 in 6 in Mercury (sg = 13.54)
-
The Wall Street Journal reported interest rates of 6 percent, 6.35 percent, 6.65 percent, and 6.75 percent for three-year, four-year, five-year, and six-year Treasury notes, respectively. According...
-
Lu Ltd. and Zha`o Ltd., two corporations of roughly the same size, are both involved in the manufacture of in-line skates. Each company depreciates its plant assets using the straight-line approach....
-
Rank the following gases from least dense to most dense at 1.00 atm and 298 K: SO 2 , HBr, CO 2 . Explain.
-
Many companies today choose to outsource even key elements of product de- sign and development. PEP designs is a leading supplier of outsourced engineering and manufacturing product development for...
-
On January 1, 2012, Valuation Allowance for Trading Investments had a zero balance. On December 31, 2012, the cost of the trading securities portfolio was $ 105,800 and the fair value was $101,600....
-
Biotech sold a patent on a new blood analyzer to Pharma. The sales agreement which was signed on January 1, 2019, requires Pharma to pay Biotech $1 million immediately. In addition, Pharma is...
-
Austin Co. established a petty cash fund by issuing a check for $300 and appointing Steve Mack as petty cash custodian. Mack had vouchers for the following petty cash payments during the month:...
-
Figure 3.33 shows a manometer being used to indicate the difference in pressure between two points in a pipe. Calculate (p A - p B ). Oil (sg = 0.90) 3 ft 2'ft 6 ft Water
-
a. Determine the gage pressure at point A in Fig. 3.36. b. If the barometric pressure is 737 mm of mercury, express the pressure at point A in kPa(abs). Water 215 mm Mercury (sg = 13.54) 600 mm
-
Which is the same for a 10- and a 20- resistor in series in a series circuit: current or voltage?
-
As a financial Manager, how will you explain the concept of Risk and Return to a potential investor, using two companies of your choice, demonstrate to the potential investor to make an informed...
-
(10) A 15uF capacitor is fully charged with a 20V battery. a) What is the charge on the capacitor? b) The same capacitor is still connected to the same battery as above, but now you insert a...
-
Most nations elect their national leader using a process known as the National Popular Vote while the United States uses the Electoral College. What are the pros and cons of the Electoral College,...
-
1. You are looking at two borrowing options. Option A charges 5%, with daily compounding. Option B charges 5.1% with semiannual compounding. Which one would you choose?Why?
-
Provide the analysis of the kinetic energy of the massive vertical spring of Length L and mass, demonstrating that the motion of its end identical with the ideal massless vertical spring having mass...
-
Find the value of the expression 4x 3 + 3x 2 x + 2 if x = 1. What is the value if x = 2?
-
d) For die casting processes: 1. What are the most common metals processed using die casting and discuss why other metals are not commonly die casted? 2. Which die casting machines usually have a...
-
A 1.50 mole sample of an ideal gas at 28.5C expands isothermally from an initial volume of 22.5 dm 3 to a final volume of 75.5 dm 3 . Calculate w for this process a. For expansion against a constant...
-
Derive the equation (H/T) V = C V + V/ from basic equations and definitions.
-
Consider the reaction TiO 2 (s) + 2 C(graphite) + 2 Cl 2 (g) 2 CO(g) + TiCl 4 (l) for which ÎH o R ,298 K = 80. kJ mol 1 . Given the following data at 25°C, Assume that the heat capacities...
-
Seved Required information [The following information applies to the questions displayed below.] On December 1, Jasmin Ernst organized Ernst Consulting. On December 3, the owner contributed $83,220...
-
For Sandhill Company, variable costs are 70% of sales and fixed costs are $191,100. Calculate the required sales in dollars that are needed to achieve management's target operating income of $73,500....
-
Japan Company produces lamps that require 2 standard hours per unit at a standard hourly rate of $18.30 per hour. Production of 7,900 units required 15,330 hours an hourly rate of $18.70 per hour....

Study smarter with the SolutionInn App


