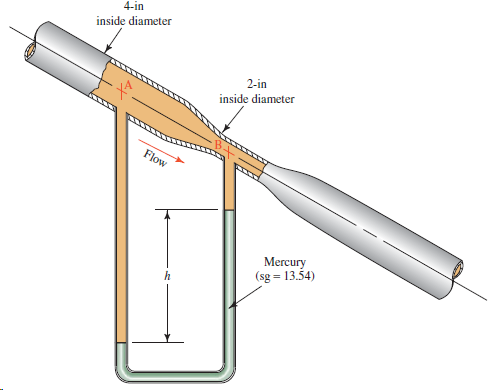
Oil with a specific gravity of 0.90 is flowing downward through the venturi meter shown in Fig.
Question:
Transcribed Image Text:
4-in inside diameter 2-in inside diameter Flow Mercury (sg = 13.54)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
v A v B 4 100 fts4 2...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A closed tank contains oil with a specific gravity 0.85. If the gauge pressure at a point 10 feet below the oil surface is 23. 7 psi (lb/in.2), determine the absolute pressure and gauge pressure (in...
-
A horizontal pipe carries oil with a specific gravity of 0.83. If two pressure gages along the pipe read 74.6 psig and 62.2 psig, respectively, calculate the energy loss between the two gages.
-
The venturi meter shown in Fig. 6.32 carries oil (sg = 0.90). The specific gravity of the gage fluid in the manometer is 1.40. Calculate the volume flow rate of oil. 75-mm inside diameter Flow 0.25 m...
-
Brennan Physiotherapies had a beta of 0.85. Reasonable estimates for the RF and the required rate of return on the market, R(Rm) were 7% and 15%, respectively. What is the required rate of return on...
-
Suppose Lululime uses the perpetual inventory system and purchases $160,000 of women's sportswear on account from Spanner Inc. on August 1, 2017. Credit terms are 2/10, net 30. Lululime pays...
-
Presented below are two independent situations: (a) On January 1, 2015, Excess Inc. purchased undeveloped land that had an assessed value of $261,000 at the time of purchase. A $500,000,...
-
Clarmont Resources has the following account balances at October 31, 2010. The inventory balance was determined using FIFO. Clarmont Resources has determined that the replacement cost (current market...
-
On January 1, 2010, Furball Company had Accounts Receivable $98,000 and Allowance for Doubtful Accounts $8,100. Furball Company prepares financial statements annually. During the year the following...
-
The following information pertains to Courvoisier Company: Month Sales Purchases January $40,000 $26,000 February $30,000 $30,000 March $60,000 $48,000 The company's sales are 25% cash and 75%...
-
Refer to information in Exercises 19-6 and 19-7. Set up T-accounts for each of the following accounts, each of which started the month with a zero balance: Raw Materials Inventory, Work in Process...
-
Draw a plot of elevation head, pressure head, velocity head, and total head for the siphon system shown in Fig. 6. 27 and analyzed in Problem 6.72. Problem 6.72 For siphon shown in Fig. 6.27,...
-
Oil with a specific gravity of 0.90 is flowing downward through the venturi meter shown in Fig. 6.33. If the manometer deflection h is 28 in, calculate the volume flow rate of oil. 4-in inside...
-
Why may a governments reported pension expenditure differ from its annual pension cost?
-
Consider the two one-shot investment alternatives shown in the table below. Neither alternative is expected to be available again in the future. MARR is 11 percent/year. Based on a present worth...
-
Enter up the Sales Day Book from the following details. Post the items to the relevant accounts in the Sales Ledger and then show the transfer to the sales account in the General Ledger. 2017 January...
-
The following sales have been made by W. Steel Ltd during the month of March 2016. All the figures are shown net after deducting trade discount, but before adding VAT at the rate of 10 per cent. You...
-
Show that if you have a mass \(M\) distributed of on a spherical shell, and a point mass \(m\) lies inside from the sphere, the gravitational potential energy is \(V=-G \frac{m M}{R}\), that is,...
-
If a local density operator is expressed by \(ho(r)=\sum_{i} \delta\left(r-r_{i} ight)\), where the sum is over particles and \(\delta\left(r-r_{i} ight)\) is the Dirac delta function, what is its...
-
Which type of constraint should you use to ensure that every book has a profit margin between 15% and 25%?
-
A container holds 2.0 mol of gas. The total average kinetic energy of the gas molecules in the container is equal to the kinetic energy of an 8.0 10-3-kg bullet with a speed of 770 m/s. What is the...
-
The mass spectrum of 2-bromopentane shows many fragments. a) One fragment appears at M79. Would you expect a signal at M77 that is equal in height to the M79 peak? Explain. b) A fragment appears at...
-
When treated with a strong base, 2-bromo-2,3-dimethylbutane will undergo an elimination reaction to produce two products. The choice of base (ethoxide vs. tert-butoxide) will determine which of the...
-
Propose a molecular formula that fits the following data. a) A hydrocarbon (C x H y ) with a molecular ion peak at m/z = 66 b) A compound that absorbs IR radiation at 1720 cm -1 and exhibits a...
-
SFS Corporation is considering opening fast food outlets in major metropolitan areas. The target leverage ratio (D/V) for this enterprise is 0.23. SFS has identified two companies in the fast-food...
-
Name the seven diatomic elements in order ofincreasing Z. hydrogen, nitrogen, fluorine, oxygen, iodine,chlorine, bromine Identify the name for each polyatomic ionshown. CO 3 2- , ClO 4 - , PO 4 3- ,...
-
Malt Ltd is considering a new project that will require equipment costing $2,000,000. The company believes the project will generate after-tax cash flows of $350,000 per year forever, with the first...

Study smarter with the SolutionInn App


