Refer to Fig. 4.40. 8,0 in Semicircular hatch 10 in 26 in 20-in radius Ethylene glycol sg
Question:
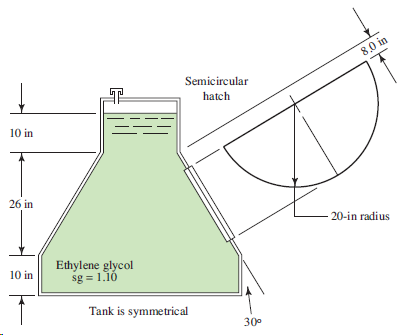
Transcribed Image Text:
8,0 in Semicircular hatch 10 in 26 in 20-in radius Ethylene glycol sg = 1.10 10 in Tank is symmetrical 30°
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a 10 incos 30 1155 in L c a 8 y 1155 8...View the full answer

Answered By

Emel Khan
I have the ability to effectively communicate and demonstrate concepts to students. Through my practical application of the subject required, I am able to provide real-world examples and clarify complex ideas. This helps students to better understand and retain the information, leading to improved performance and confidence in their abilities. Additionally, my hands-on approach allows for interactive lessons and personalized instruction, catering to the individual needs and learning styles of each student.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Refer to Fig. 4.33. Water 8 ft Access hatch for cleaning 30 in 3 ft 45 18 in 18 in 18 in
-
Refer to Fig. 4.34. 6-in diameter 3 ft 2 ft 1 ft View port Oil (sg -0.90) 30
-
Refer to Fig. 4.35. 0.6 m Oil (sg - 0.90) +300 Window 0,6 m dimen sions in mm 0.3 m 40 -1.2 m + 300+300
-
Stock Valuation While it comes to investing, picking the right stock plays a crucial role. Careful research and patience are needed. One needs to check several aspects of the company before...
-
Hecuba Company's internal controls over cash disbursements provide for the treasurer to sign checks imprinted by a check-writing machine in indelible ink after comparing the check with the approved...
-
Peter and Reesa can book their Horizon Holiday package at the early-booking price of $3900, or wait 4 months and pay the full price of $3995. a. Which option should they select if money can earn a...
-
Samano Company uses flexible budgets to control its selling expenses. Monthly sales are expected to range from \($170,000\) to \($200,000\). Variable costs and their percentage relationship to sales...
-
Hammer Orthopedic Corporation periodically invests large sums in marketable equity securities. The investment policy is established by the investment committee of the board of directors and the...
-
Marcus, a resident of Austin, Texas, has found the ideal gift for his wife in celebration of their upcoming wedding anniversarya $22,000 diamond tennis bracelet. However, Marcus is appalled at the...
-
For this assignment you will be creating a Python script to lookup phone numbers in a data file given a user-provided last name, or a first and last name. The script should look up all entries in the...
-
Refer to Fig. 4.39. 0.80 m 05 m Semicircular hatch 1.50-m diameter Turpentine 70 sg - 0.88
-
What is an SQL injection attack and how can it be prevented?
-
The density of mercury is 13,600 kg/m 3 . What is the speed of sound in mercury at 20C?
-
Upon joining a newly set-up company based in Malaysia, your have been assigned to lead a team to purchase an aircraft where you are required to fulfill mandatory requirements as required by the...
-
It has come to the attention of the National Payroll Institute Academic Integrity Committee that your assignment has been flagged with a possible violation. We require further clarification in...
-
Discuss the Chinese governments influence on the value of the yuan and how this is affecting the U.S. economy. In today's economic environment, which factor appears to have the greatest influence on...
-
The budgeted sales for the four months of operation are provided below : The budgeted sales and sales in units are 1.September-470000and 570 2.October-500000and 600 3.November-650000and 750...
-
IDK just paid a dividend of $3.20 and is expected to increase future dividends at 4% per year forever.If the current share price (V) is $34.50 what is the return required by shareholders?Remember to...
-
Use U universal set = {0, 1, 2, 3, 4, 5, 6, 7, 8, 9}, A = {1, 3, 4, 5, 9}, and C = {1,3,4,6} to find the set. (A B)
-
Consider the combustion of methanol below. If 64 grams of methanol reacts with 160 grams of oxygen, what is the CHANGE in volume at STP. 2CH3OH(g) + 3O2(g) 2CO2(g) + 4H2O(1) The volume decreases by...
-
Consider the following two compounds. When treated with NaOH, one of these compounds forms an epoxide quite rapidly, while the other forms an epoxide very slowly. Identify which compound reacts more...
-
One mole of Ar initially at 310. K undergoes an adiabatic expansion against a pressure P external = 0 from a volume of 8.5 L to a volume of 82.0 L. Calculate the final temperature using the ideal gas...
-
Predict the products for each of the following reactions: a. b. c. d. . Ti[OCH(CH,),1. (+)-DET -- THCICH) (-)-DET
-
hello , can you please help me answer those a question correctly, thank you. Saved Help Save & Exit Subn Keisha owns 100% of DEF Company and has a basis ofof $12,000. Keisha has a: $10,000 in its...
-
Accountants that are focused on issuing financial statements to serve the dicision making needs of external users of the business are called
-
Under the default provisions, the financial rights between member-management owners and manager-management owners is: Question 2 options: a) immaterial. b) material, giving a higher percentage of...
Crowdfunding Collective Investment Exponential Return 1st Edition - ISBN: 979-8859713950 - Free Book

Study smarter with the SolutionInn App


