Table 15.2 shows the proton and nucleon numbers of several nuclei. Determine the number of neutrons in
Question:
Table 15.2 shows the proton and nucleon numbers of several nuclei. Determine the number of neutrons in the nuclei of the following elements shown in the table:
a. Nitrogen
b. Bromine
c. Silver
d. Gold
e. Mercury.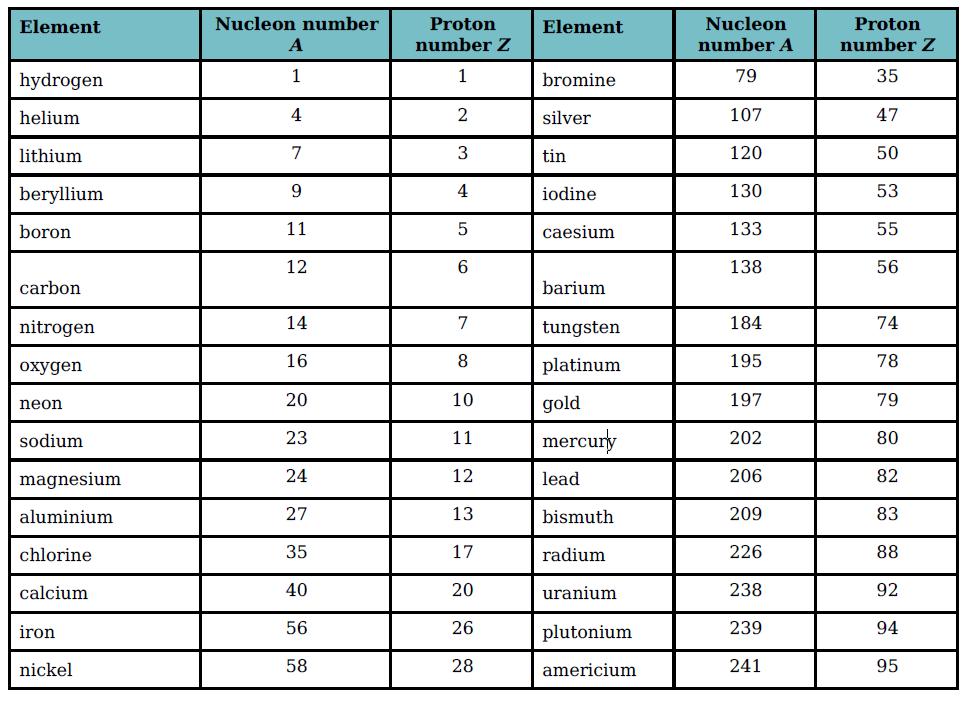
Transcribed Image Text:
Element Nucleon number Proton Element Nucleon Proton number Z number A number Z hydrogen 1 1 bromine 79 35 helium 4 2 silver 107 47 lithium 7 3 tin 120 50 beryllium 9 iodine 130 53 boron 11 caesium 133 55 12 138 56 carbon barium nitrogen 14 tungsten 184 74 oxygen 16 8 platinum 195 78 20 10 gold 197 79 neon mercury sodium 23 202 80 magnesium 24 12 lead 206 82 aluminium 27 13 bismuth 209 83 chlorine 35 17 radium 226 88 calcium 40 20 uranium 238 92 iron 56 26 plutonium 239 94 nickel 58 28 americium 241 95
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
Nitrogen has 7 protons and 7 neutrons b Bromine has 35 protons and 45 neutrons c Silver has 47 p...View the full answer

Answered By

Akshay Shete
I have extensive experience as a tutor, both online and in-person. I have worked with students of all ages and abilities, and am skilled at adapting my teaching style to meet the needs of each individual student. I have a strong background in a variety of subjects, including math, science, and English, and am able to break down complex concepts in a way that is easy for students to understand. In addition to my subject matter expertise, I am also a patient and supportive teacher, and am committed to helping my students succeed. Whether I am working with a struggling student who needs extra help to catch up, or an advanced student looking to get ahead, I am able to provide the guidance and support they need to reach their goals. Overall, my hands-on experience as a tutor has prepared me to be a confident and effective teacher, and I am excited to use my skills to help students succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Physics Coursebook
ISBN: 9781108859035
3rd Edition
Authors: David Sang, Graham Jones, Gurinder Chadha, Richard Woodside
Question Posted:
Students also viewed these Sciences questions
-
The following table gives numbers of electrons, protons, and neutrons in atoms or ions of a number of elements. Answer the following: (a) Which of the species are neutral? (b) Which are negatively...
-
Proton in a well figure shows electric potential V along an x axis. The scale of the vertical axis is set by Vs = 10.0 V. A proton is to be released at x = 3.5 cm with initial kinetic energy 4.00eV....
-
In bombarding atomic nuclei with proton "bullets," why must the protons be accelerated to high energies if they are to make contact with the target nuclei?
-
Revvit is an online motorcycle equipment store that sells motorcycle helmets, boots, jackets, and more. With no brick-and-mortar stores, Revvit relies on many systems to provide an excellent customer...
-
To expand his transmission shop, Hans needs to save $14 000 for new equipment. How much would he have to pay into an account at the beginning of every three months over three years if interest is 7%...
-
For the state of stress shown in Fig. P-931. determine normal and shearing stresses on the planes whose normals are at +60 and +150 with the x axis, Show these stresses on a sketch of the elemeni. 8...
-
A company reports the following beginning inventory and purchases, and it ends the period with 30 units in inventory. a. Compute ending inventory using the FIFO periodic system. b. Compute cost of...
-
Describe the layout of a typical fast-food franchise such as McDonalds. What type of layout is it? How does it support productivity? Do different franchises (e.g., Burger King or Wendys) have...
-
L Does JM bisect
-
ABC Company produces a chemical. At the start of the year, they had the following cost information: Direct material: (10 pounds @ $1.60) $16.00 Direct labor: (0.75 hours @ $18.00) $13.50 Variable...
-
Explain why the most strongly ionising radiation (-particles) is the least penetrating, while the least ionising (-rays) is the most penetrating.
-
Before Rutherfords model, scientists believed that the atom was made up of negatively charged electrons embedded in a plum pudding of positive charge that was spread throughout the atom. Explain how...
-
The Albring Company sells electronics equipment, and has grown rapidly in the last year by adding new customers. The audit partner has asked you to evaluate the allowance for doubtful accounts at...
-
Financial leverage Max Small has outstanding school loans that require a monthly payment of $1,080. He needs to buy a new car for work and estimates that this purchase will add $346 per month to his...
-
Provide explanation of each transaction, and in ste Indicate the effect of each of the following transactions on assets, liabilities, and owner's equity by inserting ??+? for increase and ??-? for...
-
China's huge supply of workers and it's labor force will peak around 802m in 2013-2018 has been a boon for low-cost manufacturers and has kept wages low. China also has other factors that bring down...
-
After XYZ Corporation terminated Tina's employment, Tina brought suit claiming that XYZ breached the terms of its employee handbook by failing to identify any violations of the provisions set forth...
-
The Acme Company buys 3 0 0 units of merchandise in January at $ 5 each. In February, Acme buys 5 0 0 units at $ 4 each and in March it buys 2 0 0 units at $ 6 each. Acme sells 1 5 0 units during...
-
Penmark Orchards and Cannery is considering buying high pressure cleaning equipment that vacuums waste liquids into a storage tank and then purifies the waste. The waste water can then be pumped back...
-
If |62x|>9, which of the following is a possible value of x? A. 2 B. 1 C. 0 D. 4 E. 7
-
Each of the following mechanisms contains one or more errorsthat is, the curved arrows may or may not be correct. In each case, identify the errors and then describe what modification would be...
-
In an intramolecular proton transfer reaction, the acidic site and the basic site are tethered to the same molecule, and a proton is passed from the acidic region of the molecule to the basic region...
-
For each pair of compounds below, identify the more acidic compound: (a) (b) (c) (d) (e) (f) (b) C (d) H. -S -3- (f) X H' `H.
-
Assignment A partnership is a legal entity formed by two or more people who agree to share the profits and losses of a business. Partnership accounting is the process ofrecording and reporting the...
-
Which statement below correctly describes merchandise inventory?Multiple choice question.Merchandise inventory is an asset reported on the balance sheet and contains the cost of products purchased...
-
Consulta las fuentes orientadas y otras de inters relacionadas con la asignatura y realiza las siguientes actividades: Con los conocimientos adquiridos en la unidad y las orientaciones del...

Study smarter with the SolutionInn App


