The p.d. V across a wire of length l is given by the formula V = 4IpI/d
Question:
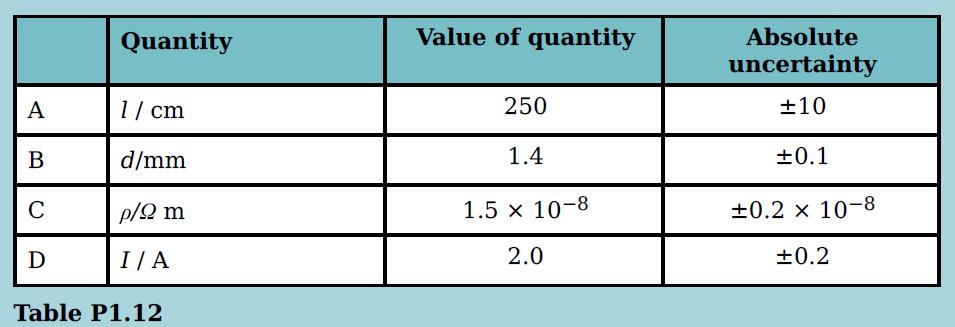
The p.d. V across a wire of length l is given by the formula V = 4IpI/d2 where d is the diameter of the wire, ρ is the resistivity and there is a current I in the wire. Which quantity provides the largest contribution to the percentage uncertainty in V?
Transcribed Image Text:
Quantity Value of quantity Absolute uncertainty А 1/ cm 250 ±10 В d/mm 1.4 ±0.1 C p/2 m 1.5 x 10-8 +0.2 x 10-8 D I/ A 2.0 +0.2 Table P1.12
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
B Because B provides the largest contribution to the percentage uncertainty in V bec...View the full answer

Answered By

Nikka Ella Clavecillas Udaundo
I am a hard-working individual with vast experience in essay, blog, and article writing. I possess skills in network configuration, troubleshooting and managing it, research, content writing(tech, academic, and marketing) and web testing skills. I provide quality work with 100% accuracy, efficiency, and delivery within the stipulated timeline
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Physics Coursebook
ISBN: 9781108859035
3rd Edition
Authors: David Sang, Graham Jones, Gurinder Chadha, Richard Woodside
Question Posted:
Students also viewed these Sciences questions
-
A voltage V is connected across a wire of length and radius r. How is the electron drift speed affected if (a) is doubled, (b) r is doubled, (c) V is doubled, assuming in each case that other...
-
A 13.0 g wire of length L = 62.0 cm is suspended by a pair of flexible leads in a uniform magnetic field of magnitude 0.440 T (Figure). What are the (a) Magnitude and (b) Direction (left or right) of...
-
A straight copper wire of length l = 1000 m and cross-sectional area S = 1.0 mm2 carries a current I = 4.5 A. Assuming that one free electron corresponds to each copper atom, find: (a) The time it...
-
1. (9 pts) Consider a supply chain with a manufacturer and a retailer. The product is offered for a single season. Suppose the manufacturer produces at a cost of $30/unit. The manufacturer sells to...
-
Solve each of the following systems of equations. (a) 6x + 5y = 9 4x 3y = 25 (b) 12 7x = 4y 6 2y = 3x (c) 0.2a + 0.3b = 0 0.7a 0.2b = 250 (d) 4/3 b 3/5 c = -17/3 5/6 b + 4/9 c = 5/9
-
What mechanisms are available to help provide stakeholders with an assurance that a company complies with environmental regulations?
-
Although the largest errors in calculating the height of a packed column are errors in (1) mass transfer coefficients and (2) VLE data, calculation errors can also be significant because calculation...
-
Katie Pairy Fruits Inc. has a $1,000, 20-year bond outstanding with a nominal yield of 15 percent (coupon equals 15% $1,000 = $150 per year). Assume that the current market-required interest rate on...
-
(IRR of an uneven cash flow stream) Microwave Oven Programming, Inc. is considering the construction of a new plant. The plant will have an initial cash outlay of $7.4 million (= - $7.4 million), and...
-
T-accounts for the general ledger, the Raw Materials Inventory subsidiary ledger, the Work-in-Process Inventory subsidiary ledger, and the Finished Goods Inventory subsidiary ledger have been opened...
-
The position of the holes in Figure P1.5 represents attempts at measuring the position of the centre of the circle. Which one shows more random error and which shows more systematic error?
-
Look at Figure P1.5. Draw similar diagrams to represent: a. A target where the holes are both precise and accurate b. A target where the holes are neither precise nor accurate.
-
(a) What are policies? (b) Distinguish between policies and implementation. (c) Why should policies not specify implementation in detail?
-
A $100 bond with semi-annual coupons, redeemable at 105 in 8 years, can be purchased at a discount of $18.19 to yield j2 = 12%. What is the bond coupon rate, j2
-
Explanation of the concept of a culture of excellence: Identification of at least three specific organizational characteristics or practices which reflect a culture of excellence. Discussion...
-
Mr. Ram is net income is $ 70,000. Mr. Ram is married and his wife, Mia has net income of $ 8,800. Mia's 73 year old mother, Bernice, lives with them. Berinice has a mental infirmity that is not...
-
2 If A= 3-2 3+ L4 2 A3-23A 401 = 0 1 then show that 1]
-
Leroy purchased a term-to-age 65 life insurance policy at the age of 33. He turns 60 next month and is considering converting his policy to a permanent plan of insurance. When must he make this...
-
On December 31, 2017, Roker, Inc. reported notes receivable of $63,930,000. This amount represents the present value of future cash flows (both principal and interest) discounted at a rate of 11.12%...
-
Complete problem P10-21 using ASPE. Data from P10-21 Original cost ................................................................. $7,000,000 Accumulated depreciation...
-
The overlap integral for Ï g and Ï u as defined in Section 23.3 is given by Plot S ab as a function of R/a 0 for ζ = 0.8, 1.0, and 1.2. Estimate the value of R/a 0 for which S...
-
Sketch out a molecular orbital energy diagram for CO and place the electrons in the levels appropriate for the ground state. The AO ionization energies are O2s: 32.3 eV; O2p: 15.8 eV; C2s: 19.4 eV;...
-
Explain the difference in the appearance of the MOs in Problem P23.13 with those for HF. Based on the MO energies, do you expect LiH + to be stable? Do you expect LiH to be stable? * Li2s H1s 20 Lils...
-
Find the value of a 5 . 9 % Coupon Bond with a required return of 1 2 . 2 1 % and 2 0 years to maturity. Keep 2 decimal places and enter the positive numeric part only. E . g . If your calculator...
-
In a sudden shift in interest rates, within a few days of the previous question, the yield to maturity changes to 3 . 5 % . What is the new price of the bond?
-
Managing properties can be a delicate balance between keeping rents marketable and keeping up the property. Not keeping these items in balance could:

Study smarter with the SolutionInn App


