This diagram shows water in a container filled to a depth of 0.50 m. The density of
Question:
This diagram shows water in a container filled to a depth of 0.50 m. The density of water is 1000 kg m−3.
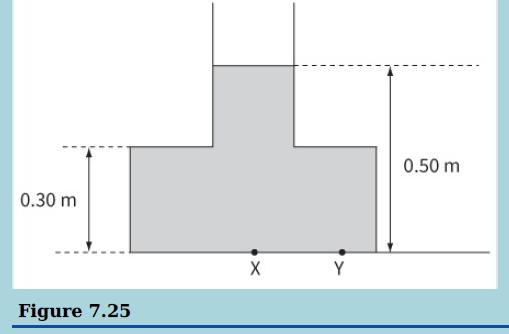
a. Calculate the pressure at X on the base of the container.
b. Explain why the pressure at X must be equal to the pressure at Y.
c. Explain why the force downwards on the base of the container is larger than the weight of the liquid in the container.
0.50 m 0.30 m Y Figure 7.25
Step by Step Answer:
a To calculate the pressure at X on the base of the container use the formula pressure density gravity height Substituting values pressure 1000 kgm3 9...View the full answer



Cambridge International AS And A Level Physics Coursebook
ISBN: 9781108859035
3rd Edition
Authors: David Sang, Graham Jones, Gurinder Chadha, Richard Woodside
Related Video
Density helps us predict whether something will float. Density is super important to consider when building things like ships and submarines. The experiment in the video examines the water density by comparing a glass of water containing sugar with a simple glass of water
Students also viewed these Sciences questions
-
Explain why values of a function must be all positive or all negative between consecutive zeros?
-
Explain why the data modeling process must be iterative. Use the Highline University example.
-
If you punch a hole in a container filled with water, in what direction does the water initially flow outward from the container?
-
Draw a topology on paper that will show the connection between home network and College (LAN) using conventional symbols used in networking? (10) Home Network: 1 connecting device 1 Wireless devices...
-
When you compare prices using internet, in which way do you use these information in your buying process?
-
If the Census did another survey, kept the error bound the same, and surveyed only 50 people instead of 200, what would happen to the level of confidence? Why? The U.S. Census Bureau conducts a study...
-
What account is debited when a partner withdraws cash for personal use?
-
Each morning, Ned Stenback stocks the drink case at Neds Beach Hut in Myrtle Beach, South Carolina. The drink case has 115 linear feet of refrigerated drink space. Each linear foot can hold either...
-
in a job order cost system, indirect labor is assogned r
-
Baker's Delight (BD) has been in the food-processing business three years. For its first two years (2005 and 2006), its sole product was raisin cake. All cakes were manufactured and packaged in...
-
Figure 7.14 shows stressstrain graphs for two materials, A and B. Use the graphs to determine the Young modulus of each material. 15- 10 0.1 0.2 0.3 0.4 Strain / % Figure 7.14: Stress-strain graphs...
-
A piece of steel wire, 200.0 cm long and having cross-sectional area of 0.50 mm 2 , is stretched by a force of 50 N. Its new length is found to be 200.1 cm. Calculate the stress and strain, and the...
-
Based on their compositions and structures and on conjugate acid-base relationships, select the stronger base in each of the following pairs: (a) BrO- or ClO-, (b) BrO- or BrO2-, (c) HPO42- or H2PO4-.
-
Find the solution set of (y-3)-(3y+4)+ 3 = 0
-
When most people think of sesame seeds, what comes to mind are those commonly found sprinkled on a hamburger bun or bagel. However, the founders of Seed+Mill had something a little different in mind....
-
Shimada Products Corporation of Japan plans to introduce a new electronic component to the market at a target selling price of $ 1 5 per unit. The company is investing $ 1 1 , 8 8 0 , 0 0 0 to...
-
How do you hash a password (hardcode the password to a hash() ) which was already created in combination of the database using php/sql? $conn = mysqli_connect("localhost: 3306", "root","",""); if...
-
Platinum alloys are used for several medical applications associated with coronary artery disease due to chemical inertness, durability and electrical conductivity. The range of platinum alloy...
-
Suppose you work for a New Zealand company exporting a container of kiwis to Haiti or Iraq. The customs official informs you that there is a delay in clearing your container through customs, and the...
-
One hundred pounds of water at atmospheric pressure are heated from 60F to 200F. What is the enthalpy change? The internal energy change? Why is the difference between the internal energy change and...
-
Why are there no points in the phase diagram for sulfur in Figure 8.11 that show rhombic and monoclinic solid phases in equilibrium with liquid and gaseous sulfur? Figure 8.11 Liquid Monoclinic-...
-
Predict which of the following substrates will undergo an E1 reaction more quickly. Explain your choice. Br Br or
-
Give an example based on moleculemolecule interactions excluding chemical reactions, illustrating how the total pressure upon mixing two real gases could be different from the sum of the partial...
-
Please provide the formulas to be used. Exchange rate is USD / EUR: 1 . 4 7 . Suppose it is end of November 2 0 0 7 , and Porsche reviews its hedging strategy for the cash flows it expects to obtain...
-
A researcher is conducting a one-way ANOVA with 3 groups, each with a sample size of 9. The MSC = 8.5 and the MSE = 4.6. What is the value of Tukey's HSD for alpha = 0.01? Select answer from the...
-
Can you please help me with question 2 part a please?

Study smarter with the SolutionInn App


