a. What do we mean by degenerate atomic orbitals? b Explain why an octahedral complex of a
Question:
a. What do we mean by degenerate atomic orbitals?
b Explain why an octahedral complex of a transition element is coloured.
c. Draw the non-degenerate 3d orbitals in a Ni2+ ion on a diagram similar to Figure 24.14. The electrons should be shown in the configuration that gives the lowest possible energy.
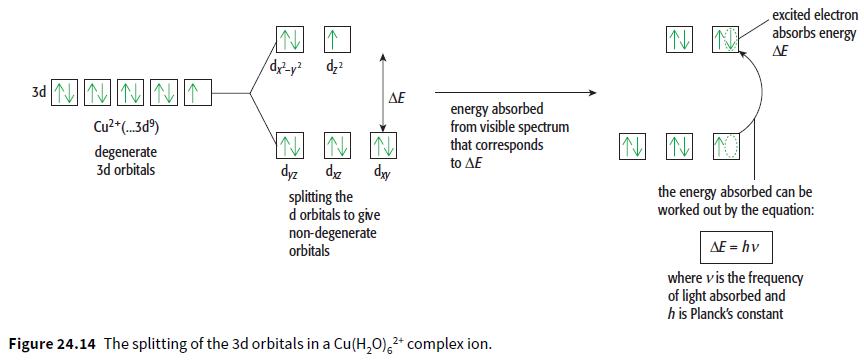
excited electron absorbs energy AE 3d N回回 AE energy absorbed from visible spectrum that corresponds Cu2 (.3d) degenerate 3d orbitals to AE dyz splitting the d orbitals to give non-degenerate orbitals dy the energy absorbed can be worked out by the equation: AF = hv where vis the frequency of light absorbed and h is Planck's constant Figure 24.14 The splitting of the 3d orbitals in a Cu(H,0),* complex ion.
Step by Step Answer:
Question a Degenerate orbitals are the orbitals of a certain subshell whic...View the full answer



Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Related Video
Using a few basic physics principles, you can impress your friends with a trick that makes a bottle disappear. For this experiment, you will need a mini plastic bottle, glycerin, a glass, and a funnel. First, open the bottle and pour some glycerin into it, then close the bottle tightly. Next, pour some water into the glass using a funnel. Place the mini bottle into the glass of water and it will look normal. This is because light travels through air faster than it travels through the glass and water, allowing our eyes to see the bottle inside the glass. However, when you fill the mini bottle with more glycerin, and pour glycerin into the glass, then put the glycerin filled bottle into the glass with glycerin in it. Half of the bottle that is submerged in the glycerin will become invisible as the light travels through glass and glycerin at the same speed, thus it does not bend and no refraction takes place, making the bottle invisible. This happens because both glass and glycerin have almost the same refractive index, which causes the speed of light to be the same in both mediums, causing no bending of light and making the bottle disappear.
Students also viewed these Sciences questions
-
What do we mean by accrued liabilities? Provide some common examples.
-
What do we mean by a progressive tax structure?
-
What do we mean by maintainable cash flows?
-
Paolo was recently promoted over Yasmine, and he now works on the sixth floor. Yasmine used to eat lunch with a coworker on the sixth floor, but lately she has been eating in the cafeteria to avoid...
-
A $5000, 14.5% bond with semi-annual coupons redeemable at par on August 1, 2026, was purchased on March 5, 2015, at 95.5. What was the approximate yield rate?
-
What are the concerns in cash management, and how do cash management ISs help financial managers?
-
Merck Pharmaceutical is a MNE with an enormous operating exposure problem. Merck does all of its product research and development and manufacturing in the United States. Because of the capital and...
-
What actions could have been taken to improve the functioning of the HP board?
-
Select the 15 Is is the seometric mean of 2 and
-
Spencer Duck (SSN 000-22-1111) is single and his eight-year-old son, Mitch, lives with him nine months of the year in a rented condominium at 321 Hickory Drive in Ames, Iowa. Mitch lives with his...
-
a. A solution of Sc 3+ ions is colourless. Suggest a reason for this. b. A solution of Zn 2+ ions is colourless. Suggest a reason for this.
-
a. Write expressions for the stability constants for the following reactions: i. [PtCl 4 ] 2 (aq) + 2NH 3 (aq) PtCl 2 (NH 3 ) 2 (aq) + 2Cl (aq) ii. [Cr(H 2 O) 6 ] 3+ (aq) + 2Cl (aq) [Cr(H 2 O)4Cl...
-
Atlantic Academy is a private school that offers education to children from Kindergarten to Grade 7. The school operates as a not-for-profit entity and oversight of the school is performed by the...
-
Describe the six progressions of management theories. Explain which management approach a manager should use and why . 1. classical viewpoint 2. Behavior viewpoint 3. Quantitative viewpoint 4....
-
How did you fare in terms of the objectives you had during your most recent performance review?
-
Straight wooden stick has mass M=0.70kg, Length L=1.6m, uniform cross-section A=14cm 2 , and constant density has a small mass m=0.20kg attached to its one end. The stick is partially submerged in...
-
Management at a newly created amusement park evaluated its customer service performance against that of Disney World s Magic Kingdom. It found gaps and used those to create a new set of best...
-
How can different laws and regulations around data privacy and security be considered when developing a communication system
-
In what ways is DCF capex analysis similar to valuing common shares, and in what ways is it different?
-
Apply Jacobis method to the given system. Take the zero vector as the initial approximation and work with four-significant-digit accuracy until two successive iterates agree within 0.001 in each...
-
Identify which two compounds shown here have the same degree of unsaturation. C 3 H 8 O C 3 H 5 ClO 2 C 3 H 5 NO 2 C 3 H 6
-
Propose all possible structures for a compound with molecular formula C 4 H 8 O that exhibits a signal at 1720 cm -1 in its IR spectrum.
-
Propose all possible structures for a compound with molecular formula C 4 H 8 O that exhibits a broad signal between 3200 and 3600 cm -1 in its IR spectrum and does not contain any signals between...
-
Your client, Li Hui Chen, has an offer to come to Canada to work as a benefits officer, specifically working in payroll administration. He has also worked as a benefits officer (NOC 13102) for the...
-
How does the state of California handle the federal educator expense deduction for tax purposes? a. California allows a full deduction, matching the federal limit of $250 for single filers and $500...
-
#3) At the beginning of 2016, Dianne Chelsey had the following loss carry forwards available: Restricted Farm Losses $7,200 Non-Capital Losses 41,000 Net Capital Losses From 2010 [(1/2)($50,000)]...

Study smarter with the SolutionInn App


