a. What is the general trend in first ionisation energies across Period 3? b. Explain why aluminium
Question:
a. What is the general trend in first ionisation energies across Period 3?
b. Explain why aluminium has a lower first ionisation energy than magnesium.
c. Explain why sulfur has a lower first ionisation energy than phosphorus.
d. Look at Period 4 in the Periodic Table. The first ionisation energies of the p-block elements are given in the table below. Predict the missing value for the first ionisation energy of selenium.
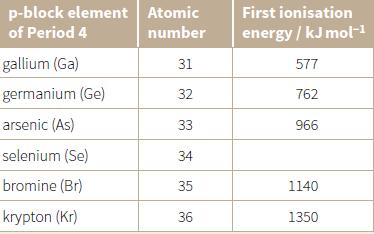
Transcribed Image Text:
p-block element Atomic First ionisation of Period 4 number energy / kJ mol-1 gallium (Ga) 31 577 germanium (Ge) 32 762 arsenic (As) 33 966 selenium (Se) 34 bromine (Br) 35 1140 krypton (Kr) 36 1350
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (9 reviews)
a The general trend in first ionisation energies across Period 3 from sodium to argon is that the fi...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Question Posted:
Students also viewed these Sciences questions
-
a. Look at Figure 11.3. i. What is the general trend in the melting points going down Group 2? ii. Which element breaks the trend? b. Explain why the atoms in Group 2, as in any other group, get...
-
What is the general relationship between mortgage rates and long-term government security rates? Explain how mortgage lenders can be affected by interest rate movements. Also explain how they can...
-
What is the general relationship among operating leverage, financial leverage, and the total leverage of the firm? Do these types of leverage complement one another? Why or why not?
-
A line charge density pL of length L lies parallel to an infinite sheet of surface charge density ps. How much work is required to rotate the line charge so that it is vertical?
-
Professor Stone complains that student teacher ratings depend on the grade the student receives. In other words, according to Professor Stone, a teacher who gives good grades gets good ratings, and a...
-
If the Marifield Steel Fabrication Company earned $500,000 in net income and paid a cash dividend of $300,000 to its stockholders, what are the firms earnings per share if the firm has 100,000 shares...
-
Core Petroleum started its oil and gas exploration and production business in 2015. During the years 2015 and 2016, the company provided the following information relating to leases located both in...
-
The preliminary 2018 income statement of Alexian Systems, Inc., is presented below: ___________________________________ALEXIAN SYSTEMS, INC. _______________________________________Income Statement...
-
5325&29 AM Question The figure below is a triangular prism. T-2.5 Geometry-3d (A) 18 cm What is the volume of the prism if the area of its base is 14 square centimeters? A B $2 cubic centimeters 126...
-
First Solar, Inc., adopted the new revenue recognition standard, ASC Topic 606, in 2017. The following are condensed versions of First Solars balance sheet, income statement, and cash flow statement,...
-
a i Describe how the atomic radius varies across Periods 2 and 3. ii. Explain this trend. b. i. Describe how the atomic radius varies down each group of the Periodic Table. ii. Explain this trend.
-
a. i. The Group 1 metal lithium reacts in a similar way to sodium. It reacts with oxygen, producing lithium oxide. Write the balanced symbol equation, including state symbols, for this reaction. ii....
-
Gumball Candies manufactures jaw- breaker candies in a fully automated process. The company recently purchased a machine that can produce 4,500 candies per month. The machine costs $ 8,000 and is...
-
You are the Head of IT Security at a financial institution in Malaysia. Due to the rise in cyber crime in recent years, you have been asked to prepare a plan of action to combat phishing (for online...
-
Case 9.1 Am I Really a Leader? Sally Helgesen was born in the small Midwest town of Saint Cloud, Minnesota. Her mother was a housewife who later taught English, and her father taught speech as a...
-
In my opinion the recent controversy over government monitoring of citizens' phone and email communications is both a good thing and also a bad thing because the advent of digital surveillance...
-
Problem 4: Drugs 'R Us operates a mail-order pharmaceutical business on the West Coast. The firm receives an average of $325,000 in payments per day. On average, it takes four days for the firm to...
-
a. What does stagflation mean? b. When does stagflation occur? Use AD-AS diagram. c. How do policy-makers fight against stagflation? Give 3 examples of policy they may use for this purpose. d. What...
-
Governments often have multiple objectives in imposing a tax. In each part of this question, use a demand and supply graph to illustrate your answer. a. If the government wants to minimize the excess...
-
Refer to Example 9.15. Add the following functionality to this program: Allow the user to enter the cost of a gallon of gas on each trip and use a function, Cost() to calculate the cost of purchasing...
-
Based on your answer to Problem 17.67, propose a mechanism for the following transformation: Answer Problem 17.67 Heat CO2 heat
-
Compare the structures of 1,4-pentadiene and divinyl amine: The first compound does not absorb UV light in the region between 200 and 400 nm. The second compound does absorb light above 200 nm. Using...
-
Provide a systematic name for each of the following compounds. a. b. c. d. e. H. CH3 Br
-
Fred. Olsen es una empresa del sector servicios y primario, que combina el transporte de pasajeros con el de mercancas. "La compaa patrocina eventos deportivos en las islas. De esta forma se...
-
) Moira is considering an investment in either a corporate bond or a government bond. All of the following statements with respect to bonds and debentures are generally true; what statement is FALSE?...
-
Completa el espacio en blanco: Un informe de redes sociales efectivo proporciona contexto adicional, lo que permite que el pblico pueda _____. 1 punto validar el retorno de inversin de la campaa...

Study smarter with the SolutionInn App


