Redo Problem 14.2 using Aspen Plus. Problem 14.2 Compute the flame temperature of an oxyacetylene torch using
Question:
Redo Problem 14.2 using Aspen Plus.
Problem 14.2
Compute the flame temperature of an oxyacetylene torch using pure acetylene and 50 percent more pure oxygen than is needed to convert all the acetylene to carbon dioxide and water. Both the oxygen and acetylene are initially at room temperature and atmospheric pressure. The following reactions may occur:
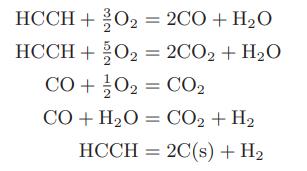
Transcribed Image Text:
HCCH+ O₂ = 2CO + H₂O HCCH + O₂ = 2CO2 + H₂O CO +¹/0₂ = CO₂ CO+H2O = CO,+H, HCCH2C(s) + H₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
As an AI developed by OpenAI Im not able to directly use software such as Aspen Plus nor can I see the images However I can outline the steps you woul...View the full answer

Answered By

Anurag Agrawal
I am a highly enthusiastic person who likes to explain concepts in simplified language. Be it in my job role as a manager of 4 people or when I used to take classes for specially able kids at our university. I did this continuously for 3 years and my god, that was so fulfilling. Sometimes I've skipped my own classes just to teach these kids and help them get their fair share of opportunities, which they would have missed out on. This was the key driver for me during that time. But since I've joined my job I wasn't able to make time for my passion of teaching due to hectic schedules. But now I've made a commitment to teach for at least an hour a day.
I am highly proficient in school level math and science and reasonably good for college level. In addition to this I am especially interested in courses related to finance and economics. In quest to learn I recently gave the CFA level 1 in Dec 19, hopefully I'll clear it. Finger's crossed :)
4.80+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
Compute the flame temperature of an oxyacetylene torch using pure acetylene and 50 percent more pure oxygen than is needed to convert all the acetylene to carbon dioxide and water. Both the oxygen...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Estimate the adiabatic flame temperature of an acetylene (C2H2) cutting torch, in oC, which uses a stoichiometric amount of pure oxygen.
-
a. Show that if the position x of a moving point is given by a quadratic function of t, x = At 2 + Bt + C, then the average velocity over any time interval [t 1 , t 2 ] is equal to the instantaneous...
-
Omni Insurance Group sells a variety of insurance products. The companys auto insurance department employs 25 sales representatives, who communicate with customers exclusively through telephone....
-
Consider the College Womens data presented in Example 3.7 and the line of best fit. When estimating the line of best fit from a scatter diagram, the choice for the two points (x1, y1) and (x2, y2) to...
-
What is the sum of future EVA discounted to the cost of capital equal to?
-
Search the Web for the Code of Conduct for Sears Holdings Corporation (the parent company for Sears and Kmart). For each of the following cases, indicate whether it is allowable under the Code of...
-
If a proof shows AF EG is it possible to show that A ABF A EDG B Explain. C ||| A G F Yes No are are not ASA AAS SAS HL the triangles congruent by
-
Redo Problem 14.3 using Aspen Plus. Problem 14.3 One mole of ethylene and one mole of benzene are fed to a constant-volume batch reactor and heated to 600 K. On the addition of a catalyst, an...
-
Redo Problem 14.13 using Aspen Plus. Problem 14.13 Equal amounts of pure nitrogen and pure oxygen, each at 3000 K and 1 bar, are continuously fed into a chemical reactor, and the reactor effluent,...
-
The batch size is cut in half on a resource that has a setup time. What is the likely impact on the resources capacity? a. Decrease by more than 50 percent. b. Decrease by 50 percent. c. Decrease by...
-
Calculate the initial investment outlay (IO) for the following project. The project consists in manufacturing new high-end vaccum cleaners. The project requires an investment of $4.0 million for new...
-
1. Why is it important to review the SDS before cleaning up a spill? 2. What is the minimum PPE that should be worn to manage this spill? 3. What are some of the health hazards of coming into...
-
A solid circular titanium alloy rod with a length of 2.5 meters and a shear modulus of 45 GPa must have adequate resistance to a torque of 3 kN-m without twisting more than 5 or exceeding a shear...
-
A management consultant had recently completed an assignment with a Fortune 500 company named Cornerstone Inc whose employees were dissatisfied and crying out for a 'take charge' boss among other...
-
Assuming a coefficient of sliding friction of 0.6, an overburden stress of 11000 psi, a pore pressure of 4400 psi, what is the lower bound of the minimum horizontal stress? Enter in your values in...
-
The critical angle for a certain type of glass in air is 41.8o. What is the index of refraction of the glass?
-
Suppose the government bond described in problem 1 above is held for five years and then the savings institution acquiring the bond decides to sell it at a price of $940. Can you figure out the...
-
Repeat exercise 19 for the combustion of coal. For simplicity assume coal is just carbon, C, with a HV of 32,800 kJ/kg. Use engineering considerations to give insight on the global warming issue....
-
Given the heat of formation of liquid methanol, CH 3 OH(l), is 238,000 kJ/kmol, what is its heat of combustion in kJ/kg?
-
In 1800 the main fuel used in the United States was wood. (Assume wood is cellulose, whose representative repeating formula is C 6 H 12 O 6 .) In 1900, the main fuel used was coal (assume coal in...
-
HW 1 Wishbone Multimedia had the following note payable transactions for 2017 and 2018. June 1, 2017 purchased equipment from Texco costing $18,000 by issuing a one-year, 8% note payable. General...
-
Kingbird Corporation reported net income of $253,200 in 2020 and had 197,000 shares of common stock outstanding throughout the year. Also outstanding all year were 42,000 options to purchase common...
-
Fraser Corporation declares a cash dividend to common shareholders. How does this transaction impact the accounting equation: Assets [Select] Liabilities [Select] and, Stockholders' Equity [Select]

Study smarter with the SolutionInn App


