Butadiene reacts to form its dimer according to the equation 2C 4 H 6 (g) C
Question:
Butadiene reacts to form its dimer according to the equation 2C4H6(g) → C8H12(g) The following data were collected for this reaction at a given temperature:
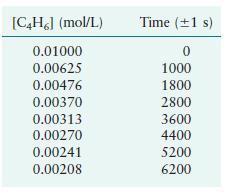
a. Is this reaction first order or second order?
b. What is the value of the rate constant for the reaction?
c. What is the half-life for the reaction under the conditions of this experiment?
Transcribed Image Text:
[C4H6] (mol/L) 0.01000 0.00625 0.00476 0.00370 0.00313 0.00270 0.00241 0.00208 Time (1 s) 0 1000 1800 2800 3600 4400 5200 6200
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
Figure 155 a To decide whether the rate law for this reaction is first o...View the full answer

Answered By

Vikash Gupta
I am graduated in Physics in 2018, from KIRORIMAL COLLEGE, University of Delhi. Now I am persuing Master's degree in physics. I like to do physics problems. I have experience of 1 year in tutoring. I think Physics is the only subject where you understand things,how they are happening . In physics you learn Maths and apply it. So I would like to join your platform to solve many Physics problems.
5.00+
5+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The tabulated data were collected for this reaction at a certain temperature: a. Determine the order of the reaction and the value of the rate constant at this temperature. b. What is the half-life...
-
The following data were collected for the rate of disappearance of NO in the reaction 2 NO(g) + O2(g) -- 2 NO2(g): (a) What is the rate law for the reaction? (b) What are the units of the rate...
-
The following data were collected in two studies of the reaction 2A + B C + D where In experiment 1, [B] 0 = 5.0 M. In experiment 2, [B] 0 = 10.0 M. a. Why is [B] much greater than [A]? b. Give the...
-
Consider the integral I = f(x) da where f(x) is the improper rational function (i) Use long division to rewrite f as the sum of a regular polynomial and a proper rational function. (ii) Factorise the...
-
Solve the equation y = xx2 + 1 / (ye y) and graph several members of the family of solutions (if your CAS does implicit plots). How does the solution curve change as the constant C varies?
-
A simple electric circuit consisting of a resistor, a capacitor, and an inductor is depicted in Fig. P2.6. The charge on the capacitor q(t) as a function of time can be computed as where t = time, q...
-
Prior to the Tax Cuts and Jobs Act of 2018 (TCJA), multinational firms domiciled in the United States did not have to pay U.S. income taxes on foreign earnings, unless those earnings were returned...
-
On July 31, 2016, the payroll register of Reed Wholesale Company showed the following totals for the month: gross earnings, $39,600; social security tax, $2,455.20; Medicare tax, $574.20; income tax,...
-
Question 5 Explain how Div 7A operates. How is a "private company" defined for the purposes of Div 7A? Gary, an Australian resident, is the sole shareholder of MC Pty Ltd, a company incorporated in...
-
The balanced equation for the reaction of gaseous nitrogen dioxide and fluorine is The experimentally determined rate law is A suggested mechanism for this reaction is Is this an acceptable...
-
A certain first-order reaction has a half-life of 20.0 minutes. a. Calculate the rate constant for this reaction. b. How much time is required for this reaction to be 75% complete?
-
How is the amount of a fringe benefit that is taxable to an employee determined?
-
Solve only part C In March 2008, Tom Lafontaine, CEO of Avalanche Logistics, a trucking company, was evaluating a new proposal that would require substantial investment. This project was of...
-
In January, the production supervisor for Harlan, Inc. requisitioned raw materials for production as follows: Job 1 $700, Job 2 $900, Job 3 $400, and general factory use, $520. Instructions Prepare a...
-
The eye of a hurricane passes over Grand Bahama Island in a direction 60.0 north of west with a speed of 42.0 km/h. Three hours later, the course of the hurricane suddenly shifts due north, and its...
-
Factor by first looking for a greatest common factor. 36x-64
-
You have been hired to do the payroll for a small software development start - up company in Kanata, Ontario with 3 employees. This is one of several clients you have. The payroll must be done on...
-
The following list of jobs in a critical department includes estimates of their required times: a. Use the shortest operation time rule to schedule these jobs. What is the schedule? What is the mean...
-
As of January 1, 2018, Room Designs, Inc. had a balance of $9,900 in Cash, $3,500 in Common Stock, and $6,400 in Retained Earnings. These were the only accounts with balances in the ledger on January...
-
An organic compound distilled from wood was found to have a molar mass of 32.04 g mol 1 and the following composition by mass: 37.5% C, 12.6% H, and 49.9% O. (a) Write the Lewis structure of the...
-
There are three isomers of dichlorobenzene, C 6 H 4 Cl 2 , which differ in the relative positions of the chlorine atoms on the benzene ring. (a) Which of the three forms are polar? (b) Which has the...
-
Predict which of the following pairs of ions would have the greatest coulombic attraction in a solid compound: (a) Mg 2+ , S 2+ ; (b) Mg 2+ , Se 2+ ; (c) Mg 2+ , O 2+ .
-
Question 11 10 points Save Answer Tyler Trucks stock has an annual return mean and standard deviation of 12 percent and 26 percent, respectively. Michael Moped Manufacturing stock has an annual...
-
You are considering a stock investment in one of two firms (LotsofDebt, Incorporated and LotsofEquity, Incorporated), both of which operate in the same industry. LotsofDebt, Incorporated finances its...
-
A manufacturer knows that their items have a normally distributed lifespan, with a mean of 13.8 years, and standard deviation of 2 years. If 23 items are picked at random, 8% of the time their mean...

Study smarter with the SolutionInn App


