What is wrong with each of the relief configurations in Figure 913 ? Be specific. Figure 913
Question:
What is wrong with each of the relief configurations in Figure 913 ? Be specific.
Figure 913
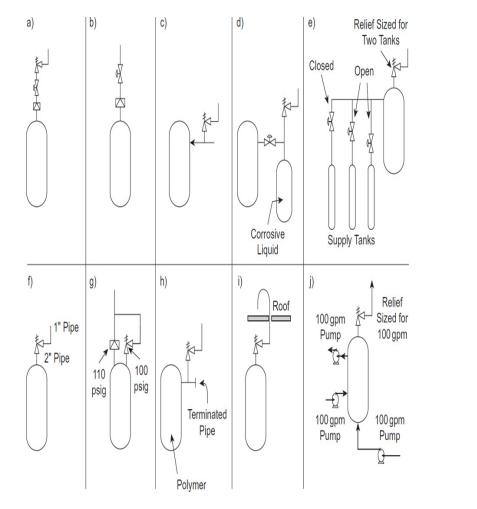
Transcribed Image Text:
a) f) 1 Pipe 2 Pipe 6 F 110 psig DR KI psig Terminated Pipe Polymer 6 i) Corrosive Liquid Roof Closed Relief Sized for Two Tanks Open -DO 100 gpm Pump 100 gpm Pump Supply Tanks DC Relief Sized for 100 gpm 100 gpm Pump
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
The image you provided shows a series of pressure relief configurations with different setups Lets discuss what might be wrong with each configuration ...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Chemical Process Safety Fundamentals With Applications
ISBN: 9780134857770
4th Edition
Authors: Daniel A. Crowl, Joseph F. Louvar
Question Posted:
Students also viewed these Engineering questions
-
Show what is wrong with each of the following electron arrangements for carbon dioxide: a. b. c. :O::O:
-
Indicate what is wrong with each of the following Lewis structures. Replace each one with a more acceptable structure. (a) Mg:0: (b) [:O-N=0:1 (c) (d) [:S-C=N:] [:a::r [C: Cl :0
-
Describe what is wrong with each of the following Lewis structures. (a) :0-C-0: (b) [.C=N:]
-
QUESTION THREE The following is an extract of a trial balance of Ruslan-Ford Partners as at 28 February 2023 Capital: Ruslan Capital: Ford Current account: Ruslan 01/03/2022 Current account: Ford...
-
A prismatically shaped gate placed at the end of a freshwater channel is supported by a pin and bracket at A and rests on a frictionless support at B. The pin is located at a distance 4 h = in. below...
-
Beckys comprehensive major medical health insurance plan at work has a deductible of $750. The policy pays 85 percent of any amount above the deductible. While on a hiking trip, she contracted a rare...
-
Identify the costs of renting.
-
Presented below are account balances for Monterey Hospital. In addition, cash transactions for the year ended December 31, 2012, are summarized in the T-account. Required: Using the information above...
-
Corporation has semiannual bonds outstanding with 14 years to maturity and the bonds are currently priced at $850. If the bonds have a coupon rate of 6 percent, then what is the after tax cost of...
-
Review Figure 9-14 and determine (a) the equipment that must be protected by a relief, (b) the relief scenarios for each relief device, and (c) the type of relief required for each location. Steam-...
-
Your company proposes to use a \(10 \mathrm{~m}^{3}\) batch reactor to react methanol + acetic anhydride in a 2:1 molar ratio. The reactor has cooling coils with a maximum cooling rate of \(30...
-
If the public expects the Fed to pursue a policy that is likely to raise short-term interest rates permanently to 5%, but the Fed does not go through with this policy change, what will happen to...
-
1) Calculate the future value of $130,000 in 40 weeks at a 5% annual interest rate. 2) Suppose you win a $300 million jackpot in a lottery, which promises to pay you $20 million per year for the next...
-
See photo attached for question Homework: MT217 M5 Competency Assessment Score: 0 of 5 pts Problem 7.LO4.16 15 of 20 (17 complete) Save HW Score: 80%, 80 of 100 pts Question Help AU.S. Government...
-
From a company's budget for next year, the operating profit before depreciation is estimated at NOK 100,000. Depreciation is estimated at NOK 20,000, while interest is estimated at NOK 10,000 and...
-
You plan on retiring in 15 years. You need $4,000 per month to live after you retire. You have average life expentency of 15 years after your retirement. How much you need to be saving every month...
-
Romance Scam are a scam artist and are looking to take advantage of a person online in an effort to defraud them of as much money as possible. In your pursuit of your criminogenic behaviour, you will...
-
Nestl S.A. is a very large company headquartered in a very small country (Switzerland). It has operations in more than 50 different countries around the world. Much of the companys international...
-
Solve the relation Exz:Solve therelation ne %3D
-
Refer to the Fourier transform infrared spectrum in Figure 19-32. (a) The interferogram was sampled at retardation intervals of 1.2660 10 -4 cm. What is the theoretical wavenumber range (0 to ?) of...
-
The table shows signal-to-noise ratios recorded in a nuclear magnetic resonance experiment. Construct graphs of (a) signal-tonoise ratio versus n and (b) signal-to-noise ratio versus n, where n is...
-
Would you use a tungsten or a deuterium lamp as a source of 300-nm radiation? What kind of lamp provides radiation at 4-m wavelength?
-
The trial balance for December 31, 2021, included the following accounts: Account Title Sales revenue Cost of goods sold Selling and administrative expense Interest expense Gain on debt securities...
-
I am unclear on how to get the flexible budget. . Osage, Inc_, manufactures and sells lamps. The company produces only when it receives orders and, therefore, has no inventories. The following...
-
. Question 2 (Q2-a) In a pharmaceutical research company, Sarah was a highly skilled chemist who had been working on a groundbreaking drug discovery project. She had dedicated countless hours to...

Study smarter with the SolutionInn App


