An electrochemical cell is based on these two half-reactions: Calculate the cell potential at 25 C. 2+
Question:
An electrochemical cell is based on these two half-reactions: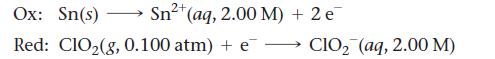
Calculate the cell potential at 25 °C.
Transcribed Image Text:
2+ Sn²+ (aq, 2.00 M) + 2 e Ox: Sn(s) Red: ClO₂(g, 0.100 atm) + e- CIO₂ (aq, 2.00 M)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Sure to calculate the cell potential Ecell for the electrochemical cell you can use the ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
An electrochemical cell is based on these two half-reactions: Calculate the cell potential at 25 C. Ox: Pb(s) Pb+ (aq, 0.10 M) + 2 e Red: MnO4 (aq, 1.50 M) + 4H* (aq, 2.0 M) + 3 e MnO (s) + 2 HO(1)
-
An electrochemical cell is constructed such that on one side a pure nickel electrode is in contact with a solution containing Ni2+ ions at a concentration of 3 10-3 M. The other cell half consists...
-
The reaction taking place in an electrochemical cell under standard conditions is Fe 2+ (aq) + Ag + (aq) Fe 3+ (aq) + Ag(s) a. Write two half-equations for this reaction. For each, state whether...
-
A UK company has a trading loss of 50,000 for the year to 31 March 2021. During the year, the company receives overseas property income (net of 40% withholding tax) of 12,000. Show the corporation...
-
1. A unit-level driver is consumed by a product each and every time that a. a batch of products is produced. b. a purchase order is issued. c. a unit is produced. d. a customer complains. e. none of...
-
You ask for a students age at his or her last birthday. Determine the number of outcomes in each event. Then decide whether each event is simple or not. Explain your reasoning. 1. Event C: The...
-
Division A of Incorrigible Dysfunctionals Limited is the only source of supply for an intermediate product that is converted by Division B into its final saleable form. Most of A's costs are fixed....
-
Lamb Inc. produces calendars in a two-process, two-department operation. In the Printing Department, calendars are printed and cut. In the Assembly Department, the material received from Printingis...
-
Question 4 (2 POINTS) Law of Cosines At an airport radar tower, the air traffic controller was able to determine how far two planes were away from the airport and the included angle. How far were the...
-
What is a concentration electrochemical cell?
-
Explain the difference between a voltaic (or galvanic) electrochemical cell and an electrolytic cell.
-
The rod has a length L and mass m.A smooth collar having a negligible size and one-fourth the mass of the rod is placed on the rod at its midpoint. If the rod is freely rotating at about its end and...
-
The position of an object as a function of time is given as x = At Bt2 + Ct + D. The constants are A = 2.21 m/s, B = 1.31 m/s, C= -4.85 m/s, and D = 3.25 m. Problem 02.037.c - Position of an object...
-
As shown below, a box slides on a flat horizontal surface towards a spring. When the box hits the spring, it compresses the spring a distance x and finally is brought to rest (momentarily). Use the...
-
LenDin Company uses forklifts to move heavy products. Recently one of the forklift has not functioned well and management is considering updating the forklift with a faster model. Current Printer New...
-
Light from two sources, A = 623 nm and A = 488 nm, is incident on a diffraction grating that has 5550 lines/cm. What is the angular separation, 0-0, of the second order maxima of the two waves?
-
The following information is provided for the first month of operations for Legal Services Inc.: Prepare a T-accounts to reflect the following business transaction: Legal Services, Inc. paid their...
-
Should investors be concerned only with a companys profitability? What metrics might an investor use to measure the social responsibility success of a company?
-
You are the newly appointed tax practitioner to complete Emilys tax return and have downloaded the prefill report for Emilys tax return (hint, you can read what a prefill report is here (Links to an...
-
Describe what happens to the vapor pressure of water as the temperature increases.
-
Describe why it is important to consider NPSH when designing and operating a pumping system.
-
Find the available NPSH when a pump draws water at 140F from a tank whose level is 4.8 ft below the pump inlet. The suction line losses are 2.2 lb-ft/lb and the atmospheric pressure is 14.7 psia.
-
Question 4 of 6 < > -/15 Current Attempt in Progress Sheridan Company has accounts receivable of $202,000 at September 30, 2024. An analysis of the accounts shows the following: Month of Sale Balance...
-
The charge on your rent-to-own washer is $40 per month. After 32 months, you get to keep the washer. How much does the washing machine end up costing you? Cost of washing machine
-
Prepare horizontal analysis on income statement below: Bristol Myers Squibb Co. (NYS: BMY) Report Date 2020 2019 2018 Net product sales 41,321 25,174 21,581 Alliance & other revenues 1,197 971 980...

Study smarter with the SolutionInn App


