Consider the titration curves (labeled a and b) for two weak bases, both titrated with 0.100 M
Question:
Consider the titration curves (labeled a and b) for two weak bases, both titrated with 0.100 M HCl.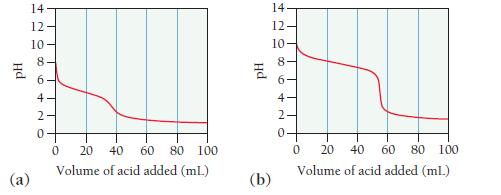
i. Which base solution is more concentrated?
ii. Which base has the larger Kb?
Transcribed Image Text:
Hd (a) 14 == 208 12 10 6 4 N 0 0 20 40 60 80 100 Volume of acid added (mL) PH (b) 14. 12- 10- 00 10 8 6- 4- 2. 0 0 20 40 60 80 100 Volume of acid added (ml.)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
The base solution in curve a is more concentrated than the base solution in curv...View the full answer

Answered By

David Ngaruiya
i am a smart worker who concentrates on the content according to my clients' specifications and requirements.
4.50+
7+ Reviews
19+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A mountain-climbing expedition establishes two intermediate camps, labeled A and B in the drawing, above the base camp. What is the magnitude (r of the displacement between camp A and camp B? 4900 m...
-
A chemical company produces amongst its product range two industrial cleaning fluids, A and B. These products are manufactured jointly. Total sales are expected to be restricted because home trade...
-
The figure in the preceding exercise shows the pH curves for the titrations of six different acids with NaOH. Make a similar plot for the titration of three different bases with 0.10 M HCl. Assume...
-
How does ESMA support national regulators in enforcing the application of IFRS?
-
What is cycle time? Velocity?
-
6P2 / 11P3 Perform the indicated calculation.
-
Although the pattern of hectic activity observed by Mintzberg is generally agreed to be a true reflection of a typical senior manager's life, it has been argued that it is a sign of a poorly...
-
The records of Eaton Aviation include the following accounts for inventory of aviation parts at July 31 of the current year: Requirements 1. Prepare a partial income statement through gross profit...
-
A graphical representation of a linear program is shown below. The shaded area represents the feasible region, and the dashed line in the middle is the objective function line. If this is a...
-
Phenolphthalein has a pK a of 9.7. It is colorless in its acid form and pink in its basic form. For each of the values of pH, calculate [In ]/[HIn] and predict the color of a phenolphthalein...
-
Calculate G and K for each reaction the group created in Question 143. For one of the reactions, explain how the sign or magnitude of each quantity (Ecell, G, and K) is consistent with the fact that...
-
A person decides to use a microwave oven to reheat some lunch. In the process, a fly accidentally flies into the microwave and lands on the outer edge of the rotating plate and remains there. If the...
-
On August 1, Giuseppe Martinez established Martinez Consulting Group. The following transactions were completed during the month. 1. Invested $25,500 cash, a new computer system and printer with a...
-
What are the potential challenges and considerations for migrating an existing monolithic relational database to a sharded architecture, and what strategies can be employed to minimize disruption and...
-
How to make a journal entry of the following: a. 10/02/20xx - Owner paid for a new vehicle with her own capital. This has increased the company assets with a capital contribution. $18,000 including...
-
Find the composition of (fog)(a) for f(x) = x-2x+3 and g(x) = x-5
-
HM has operating income (EBIT) of $750,000. The depreciation expense is $200,000. HM is 100% equity financed and faces a 40% tax rate. a. What is the company's net income? b. What is its net cash...
-
You are the Vice President of Sales for a $30 million manufacturer of home building materials. The company employs 50 salespeople around the country to market the companys products to hardware stores...
-
Find the equation of the plane passing through the points P 5,4,3 ,Q 4,3,1 and R 1,5,4
-
Figure P.11.49 shows a transparent ring on an otherwise opaque mask. Make a rough sketch of its autocorrelation function, taking l to be the center-to-center separation against which you plot that...
-
Return to Eq. (12.21) and separate it into two terms representing a coherent and an incoherent contribution, the first arising from the superposition of two coherent waves with irradiances of |? ? 12...
-
Referring to the previous two problems with the cosine grating oriented horizontally, make a sketch of the electric-field amplitude along y' with no filtering. Plot the corresponding image irradiance...
-
Cyclizar Inc. provided S4,000 of services with the customers promising to pay next week. In the journal entry written, what would go in box d? A) Cash B) Accounts Receivable C) Revenue D) Accounts...
-
ANSWER IN YOU OWN WORDS! Dont forget to provide references. Thank you. 1. Explain the process by which assets are written up (or down) on intercompany asset transfers and why GAAP prohibits the...
-
Question 4 i need answer asap The following information is given for Rise Up Sdn.Bhd: Unit sales price RM15 Variable cost per unit RM7 Total Fixed cost RM55,OOO Required: a) Calculate the...

Study smarter with the SolutionInn App


