Determine whether each redox reaction occurs spontaneously in the forward direction. a. Ca+ (aq) + Zn(s) b.
Question:
Determine whether each redox reaction occurs spontaneously in the forward direction.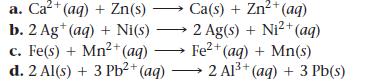
Transcribed Image Text:
a. Ca²+ (aq) + Zn(s) b. 2 Ag+ (aq) + Ni(s) c. Fe(s) + Mn²+ (aq) d. 2 Al(s) + 3 Pb²+ (aq) Ca(s) + Zn²+ (aq) 2 Ag(s) + Ni²+ (aq) Fe²+ (aq) + Mn(s) 2 Al³+ (aq) + 3 Pb(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a Ca2 aq Zns Cas Znaq The halfreactions involved are Zns Znaq 2e E 076 V Caaq 2e Cas E 287 V Since t...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Determine whether each redox reaction occurs spontaneously in the forward direction. a. Ni(s) + Zn+ (aq) b. Ni(s) + Pb+ (aq) c. Al(s) + 3 Ag+ (aq) d. Pb(s) + Mn+ (aq) 2+ Ni+(aq) + Zn(s) Ni+(aq) +...
-
At 298 K, for the reaction 2 PCl 3 (g) + O 2 (g) 2 POCl 3 (l), r H = -620.2 kJ mol -1 and the standard molar entropies, in J mol 1 K 1 , are PCl 3 (g), 311.8; O 2 (g), 205.1; and POCl 3 (l), 222.4....
-
At 298 K, for the reaction 2 H + (aq) + 2 Br - (aq) + 2 NO 2 (g) Br 2 (l) + 2 HNO 2 (aq), r H = -61.6 kJ mol -1 and the standard molar entropies are H + (aq), 0 J mol -1 K -1 ; Br - (aq), 82.4 J...
-
In a bygone day, airlines issued discount tickets to students who would be willing to fly on a particular day, with no notice, at a discounted price, one needed to show proof of being of student. The...
-
As a recently hired internal auditor for the Emerson Department Store (which has approximately 500 employees on its payroll), you are currently reviewing the stores procedures for preparing and...
-
The target population is all students in your university. You wish to estimate the average current Visa balance for each student. How large would the university student population have to be in order...
-
Peters Manufacturing Inc. operates the Home Appliance Division as a profit center. Operating data for this division for the year ended December 31, 2002, are as follows: In addition, Peters...
-
Repeat Exercise 3.31 for the FSM shown in Figure 3.73. Recall that the s and r register inputs indicate set and reset, respectively. Data From Problem 31 Analyze the FSM shown in Figure 3.72. Write...
-
Question In your own words please explain what a confidence interval is
-
Suppose you wanted to cause Ni 2+ ions to come out of solution as solid Ni. Which metal could you use to accomplish this?
-
Determine whether each reaction is a redox reaction. For each redox reaction, identify the oxidizing agent and the reducing agent. a. Al(s) + 3 Ag+ (aq) b. SO3(g) + HO(1) c. Ba(s) + Cl(g) d. Mg(s) +...
-
How does Social Security affect the economic well-being of blacks relative to whites and Hispanics? Explain.
-
Describe each speaker's verbal and non-verbal communication. Compare and contrast the strengths and weaknesses of each speaker and their communication. Explain what aspects of each speaker's verbal...
-
One Primary Source Annotation citation and annotation Share one primary source related your topic; provide the citation and a brief annotation. Just to clarify, a primary source is generally a first...
-
How is a Method different from an Object? Explain.Make sure in any explanation to use your own words and provide a comprehensive answer. Find two websites that provide specific help on Lists that you...
-
A client gives you the following feedback: "I would like to offer some feedback to you about the appraisal. You have completed the appraisal thoroughly and you have presented the information to us...
-
3 Diversification 1. Equally Weighted Portfolio (Total 20 points) (a) Setting and notations Consider an investment universe with n securities, S1, S2, ..., Sn. Given a fixed investment horizon, we...
-
Fleming, Fleming, and Johnson, a local CPA firm, provided the following data for individual returns processed for March (output is measured in number of returns): Units, beginning work in process ...
-
The Ranch 888 Noodle Company sells two types of dried noodles:ramen, at $6.50 per box, and chow fun, at $7.70 per box. So farthis year, the company has sold a total of 110,096 boxes ofnoodles,...
-
Two hockey players are traveling at velocities of v 1 = 12 m/s and v 2 = -18 m/s when they undergo a head-on collision. After the collision, they grab each other and slide away together with a...
-
Consider an elastic collision in one dimension that involves objects of mass 2.5 kg and 4.5 kg. The larger mass is initially at rest, and the smaller one has an initial velocity of 12 m/s. Find the...
-
Two billiard balls undergo an elastic collision as shown in Figure P7.27. Ball 1 is initially traveling along x with a speed of 10 m/s, and ball 2 is at rest. After the collision, ball 1 moves away...
-
77. For the company you are analyzing, the company's compensation committee recommends awarding fewer stock options to executives in the future. The company adopts this recommendation, and this is...
-
1. Which of the following is NOT authoritative guidance that applies AICPA members providing forensic accounting services? a. Generally accepted auditing standards b. AICPA Code of Professional...
-
Select all that apply Not recording an accrued expense will have the following effect on the financial statement: Multiple select question. expenses on the income statement will be overstated....

Study smarter with the SolutionInn App


