Estimate the value of the equilibrium constant at 525 K for each reaction in Problem 73. Problem
Question:
Estimate the value of the equilibrium constant at 525 K for each reaction in Problem 73.
Problem 73
Use data from Appendix IIB to calculate the equilibrium constants at 25 °C for each reaction.![]()
Appendix IIB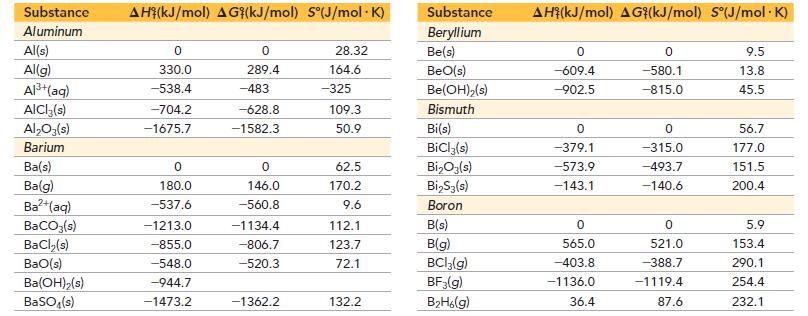
Transcribed Image Text:
a. 2 CO(g) + O₂(g) = 2 CO₂(g) b. 2 H₂S(g) = 2 H₂(g) + S₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a 190...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Estimate the value of the equilibrium constant at 655 K for each reaction in Problem 74. (H f for BrCl is 14.6 kJ/mol.) Problem 74 Use data from Appendix IIB to calculate the equilibrium constants at...
-
Use data from Appendix IIB to calculate the equilibrium constants at 25 C for each reaction. G f for BrCl(g) is -1.0 kJ/mol. Appendix IIB a. 2 NO(g) NO4(8) b. Br(g) + Cl(g) = 2 BrCI(g)
-
Use data from Appendix IIB to calculate the equilibrium constants at 25 C for each reaction. Appendix IIB a. 2 CO(g) + O(g) = 2 CO(g) b. 2 HS(g) = 2 H(g) + S(g)
-
What future directions may be interesting for comparative studies in corporate governance?
-
Explain how an activity-based budget is prepared.
-
A math conference has an attendance of 4950 people. Of these, 2110 are college professors and 2575 are female. Of the college professors, 960 are female. Find the probability that a randomly selected...
-
Define management control, and justify why you consider your definition to be an improvement on others that have been advanced.
-
A computer tape moves over the two drums shown. Drum A weighs 1.4 lb and has a radius of gyration of 0.75 in., while drum B weighs 3.5 lb and has a radius of gyration of 1.25 in. In the lower portion...
-
x- **#2.) A lighthouse is built on the edge of a cliff near the ocean, as shown in the accompanying diagram. From a boat located feet from the base of the cliff, the angle of elevation to the top of...
-
Consider the reaction: Calculate G rxn for the reaction at 25 C under each of the following conditions: a. Standard conditions b. At equilibrium c. P ICl = 2.55 atm; P I2 = 0.325 atm; P Cl2 = 0.221...
-
Describe the solubility of CaF 2 in each solution compared to its solubility in water. a. In a 0.10 M NaCl solution b. In a 0.10 M NaF solution c. In a 0.10 M HCl solution
-
A store manager reports a confidence interval of (244.07, 280.97) when estimating the mean price (in dollars) for the population of textbooks. Use the confidence interval to find the estimated margin...
-
Find the point of intersection for the 2 linear functions: x= y +6 2x-y=13
-
The original cost of an asset is 190,000 while the scrap value is expected to be 10,000. The estimated useful life of the asset is 9 years. Calculate the amount of annual depreciation using the...
-
After catching the ball, Sarah throws it back to Julie. The ball leaves Sarah\'s hand a distance 1 . 5 meters above the ground, and is moving with a speed of 8 m / s when it reaches a maximum height...
-
Balance sheet of the company Juve on 1 December 2021 ASSETS Property, plant & equipment* EQUITY & LIABILITIES 100 000 Share capital 100 000 Traded goods** 12 000 Long-term bank loan 20 000 Trade...
-
Write a static method odds () belonging to the IntLinkedBag class. odds () will have one parameter of type IntLinkedBag. The method will return an IntLinkedBag containing only the odd numbers from...
-
Many doctors do not work individually but instead are part of a group practice often connected to a local hospital. How would the sales process change for a pharmaceutical salesperson selling to a...
-
A glass manufacturer produces hand mirrors. Each mirror is supposed to meet company standards for such things as glass thickness, ability to reflect, size of handle, quality of glass, color of...
-
Redraw the ray diagram for a compound microscope (Fig. 5.110), but this time treat the intermediate image as if it were a real object. This approach should be a bit simpler. Fig. 5.110 Exit pupil fe...
-
Consider a thin positive lens L 1 , and using a ray diagram, show that if a second lens L 2 is placed at the focal point of L 1 , the magnification does not change. Thats a good reason to wear...
-
Draw a ray diagram locating the images of a point source as formed by a pair of mirrors at 90? (Fig. P.5.61a). Now create a ray diagram locating the images of the arrow shown in Fig. P.5.61b. Fig...
-
Logistics Solutions provides order fulfillment services for dot.com merchants. The company maintains warehouses that stock items carried by its dot.com clients. When a client receives an order from a...
-
Hace varios meses, la empresa Jones sufri un vertido de materiales peligrosos al ro White desde una de sus plantas. Como resultado, la Agencia de Proteccin Ambiental (EPA) mult a la empresa con...
-
On January 1, 2021, Robertson Construction leased several items of equipment under a two-year operating lease agreement from Jamison Leasing, which routinely finances equipment for other firms at an...

Study smarter with the SolutionInn App


