Use the data shown here to find the equilibrium constant (K c ) for the reaction A(g)
Question:
Use the data shown here to find the equilibrium constant (Kc) for the reaction A(g) ⇌ 2 B(g) + C(g).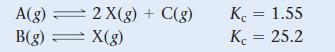

Transcribed Image Text:
A(g) = B(g) = 2X(g) + C(g) X(g) Kc = 1.55 Kc = 25.2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
d ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Determine A in the indicated figures. Fig. 2.40 (c) B 4 66 A (c) 4 C
-
The equilibrium constant Kc for the reaction equals 4.1 at 300oC. a. A sample of 35.8 g of PCl5 is placed in a 5.0-L reaction vessel and heated to 300oC. What are the equilibrium concentrations of...
-
The equilibrium constant Kc for the reaction is 3.8 Ã 10-5 at 727°C. Calculate Kc and KP for the equilibrium at the same temperature. 12(g)--21(g) 21(g)- 2(g)
-
Lead has one of the highest densities of all the pure metals. The density of lead is 11,340 kg/m. What is the density of lead in units of lbm/in?
-
Refer to Exercise 21-5. Assume the economic lot size for small casings is 30,000 and that of the large casings is 10,000. Wadley Manufacturing sells an average of 590 small casings per workday and an...
-
The 2012 income statement of Schmidt Corporation showed net income of $1,230,000 and a loss from discontinued operations of $105,000. Schmidt had 40,000 common shares outstanding all year. Prepare...
-
Sajjad Manufacturing has beginning raw materials inventor) $12.000, ending raw materials inventor) \($18,000\). and raw materials purchases $180,000. What is the 1 "siol direct materials used?
-
Prepare a master budget for McGregor Pharmacy Company for the year ending December 31, 2021 using the following information. Prepare it per quarter. Use the tables provided by the professor. Fill it....
-
S Name the x coordinate of one solution to the system of equations 2
-
Write an expression for the equilibrium constant (K c ) for this chemical equation: CaCO3(s) = CaO(s) + CO2(g)
-
Consider the reaction A(g) B(g). The images shown here illustrate equilibrium mixtures of A (red) and B (black) at three different temperatures. At which temperature is the equilibrium constant the...
-
What is the minimum dollar amount specified in the Uniform Commercial Code as a requirement for written contracts for the sale of personal property? Should this amount be greater?
-
If a charity raises money from thirty donors, and sixteen of the donors each give $20, twelve of the donors each give $50, and two of the donors each give $100, what is the weighted average donation?
-
What are the implications of habitat fragmentation, landscape connectivity, and land-use change for biodiversity conservation, and how do conservation biologists prioritize conservation efforts and...
-
How do evolutionary processes, such as speciation, extinction, and adaptive radiation, interact with ecological dynamics to shape patterns of biodiversity over geological timescales, and what...
-
How do eukaryotic cells regulate cell polarity, shape, and motility through cytoskeletal dynamics, cell-cell adhesion complexes, and membrane remodeling processes, and what are the implications of...
-
Flatland Corp. harvested potatoes at a cost of FC 1,000,000 and sold them to Pot Corp. for FC 2,500,000. Pot Corp. sold the potatoes to its customers and received FC 4,000,000. If the national VAT is...
-
An electron and a proton are moving with the same speed. (a) Compared with the proton, will the electron have (1) a shorter, (2) an equal, or (3) a longer de Broglie wave-length? Why? (b) If the...
-
The liquidliquid extractor in Figure 8.1 operates at 100F and a nominal pressure of 15 psia. For the feed and solvent flows shown, determine the number of equilibrium stages to extract 99.5% of the...
-
In the circuit in Fig. 6.64 , let i s = 4.5e 2t mA and the voltage across each capacitor is equal to zero at t = 0. Determine v 1 and v 2 and the energy stored in each capacitor for all t > 0. 36 24...
-
If v(0) = 0, find v(t), i 1 (t), and i 2 (t) in the circuit of Fig. 6.63 . Is (mA). 30 5 t -30 6 F is 4F
-
Assuming that the capacitors are initially uncharged, find v o (t) in the circuit of Fig. 6.62 . (mA) A 6 F 90 is 3 F vo(t) 2 t(s)
-
In the current tax year a taxpayer paid $6,000 to replace all 16 windows in their existing home to windows that are designed to reduce heat loss or gain. How much of the Residential Energy Home...
-
What is a good way to improve efficiency and accuracy in the registration process? a. Email audit results to staff members with instructions to improve. Training takes up too much time. b. Scan a...
-
Should Burton pursue both the thermowell machine purchase and the EE acquisition? Why or why not

Study smarter with the SolutionInn App


