Calculate the pH of the following solutions: a. 1.2 M CaBr b. 0.84 M C6H5NH3NO3 (K for
Question:
Calculate the pH of the following solutions:
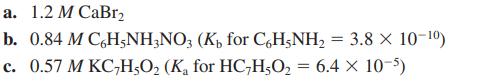
Transcribed Image Text:
a. 1.2 M CaBr₂ b. 0.84 M C6H5NH3NO3 (K₁ for C6H5NH₂ = 3.8 × 10-¹⁰) c. 0.57 M KC₂H5O₂ (K₁ for HC-H₂O₂ = 6.4 x 10-5)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Solution 1 12 M CaBr CaBr is a salt of a strong base CaOH and a strong acid HBr When dissolved in wa...View the full answer

Answered By

Arun kumar
made more than four thousand assignments
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Calculate the pH of each of the following solutions (Ka and Kb values are given in Appendix D): (a) 0.095 M propionic acid (C2H5COOH), (b) 0.100 M hydrogen chromate ion (HCrO4-, (c) 0.120 M pyridine...
-
First, write the code to create a single DataFrame object in a function called load_ticket_data(). This function should return the full dataframe and take no parameters (you can assume the ticket...
-
Pioneer Bicycle Shop is an authorized Trek dealer. In order to be an authorized Trek dealer, Pioneer and Trek signed an agreement where Trek agreed to sell Pioneer its entire line of bicycles and...
-
What are the types of production systems in aquaculture?
-
On what did the court base its reasoning for its ruling on this issue?
-
Kraft Foods Group reports the following for two of its divisions for a recent year. All numbers are in millions of dollars. For each division, compute (1) Return on investment, (2) Profit margin, (3)...
-
Metro Hospital has been under pressure to keep costs down. Indeed, the hospital administrator has been managing various revenue-producing centres to maximize contributions to the recovery of the...
-
Perfection Sound, Inc., a manufacturer of stereo speakers, is thinking about adding a new plastic-injection molding machine. This machine can produce speaker parts that the company now buys from...
-
In March, Sandhill Manufacturing had the following unit production costs: materials $14 and conversion costs $10. On March 1, it had zero work in process inventory. During March, Sandhill completed...
-
Fourth-Sixth-Twelfth Bank currently pays an annuall dividend of $1.40 per share. After extensive analysis, you forecast that the Bank's stock will continue to pay the $1.40 dividend for four years....
-
Students are often surprised to learn that organic acids, such as acetic acid, contain OOH groups. Actually, all oxyacids contain hydroxyl groups. Sulfuric acid, usually written as H 2 SO 4 , has the...
-
Calculate the pH of each of the following solutions. a. 0.12 M KNO 2 b. 0.45 M NaOCl c. 0.40 M NH 4 ClO 4
-
The flow at the end of a 30-m-long side channel spillway is 36.0 m3/sec. A 30-m-long overflow spillway, which is under a head of 0. 736 m, contributes the flow to the side-channel spillway. If the...
-
1. Roberta thinks that cycling is the best way to explore a new city. On her visit to Paris, she cycled 75 kilometers to get to her next Airbnb in the neighboring town. The town was due east of...
-
Let's prepare ourselves for the future discussions on project management in the realm of information systems. What could be the potential risk factors in information systems projects, and how can we...
-
In many locations, old abandoned stone quarries have become filled with water once excavating has been completed. While standing on a quarry wall, a boy kicks a piece of granite into the water below....
-
Consider the following. s 7 cm, r = 4 cm (a) Find the radian and degree measures of the central angle subtended by the given arc of length s on a circle of radius r. (Round the degree measure of the...
-
Two identical spaceships with proper lengths of 175 m are launched from Earth. Spaceship A is launched in one direction at 0.500c and spaceship B is launched in the opposite direction at 0.750c. a....
-
The figure shows the result of a MINITAB regression analysis of the explanatory variable x = sugar and the response variable y = sodium for the breakfast cereal data set discussed in Chapter 2 (the...
-
Gordon and Lisa estimate that they will need $1,875,000 in 40 years for their retirement years. If they can earn 8 percent annually on their funds, how much do they need to save annually?
-
Nitriles undergo alkylation at the α position much like ketones undergo alkylation at the α position. The α position of the nitrile is first deprotonated to...
-
Identify the Michael donor and Michael acceptor that could be used to prepare each of the following compounds via a Michael addition. (a) (b) (c) (d) (e) OEt N
-
The conjugate base of diethyl malonate can serve as a nucleophile to attack a wide range of electrophiles. Identify the product that is formed when the conjugate base of diethyl malonate reacts with...
-
1) The Rolling Department of Oak Ridge Steel Company had 4,800 tons in beginning work in process inventory (60% complete) on July 1. During July, 80,400 tons were completed. The ending work in...
-
Has the government refrained from including agency funds in that statement? Are increases and decreases shown as additions and deductions, rather than revenues and expenses? What are the main...
-
Accounting I12) Young Money Cash Money has net sales revenue of $793,000 cost of goods sold of$349,700 net income of $193,200, and preferred dividends of $16,500 during thecurrent year. At the...

Study smarter with the SolutionInn App


