Consider the following four titrations (iiv): a. Rank the four titrations in order of increasing pH at
Question:
Consider the following four titrations (i–iv):
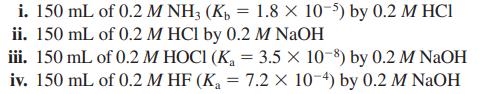
a. Rank the four titrations in order of increasing pH at the halfway point to equivalence (lowest to highest pH).
b. Rank the four titrations in order of increasing pH at the equivalence point.
c. Which titration requires the largest volume of titrant (HCl or NaOH) to reach the equivalence point?
Transcribed Image Text:
i. 150 mL of 0.2 M NH3 (K₂ = 1.8 × 10-5) by 0.2 M HCI ii. 150 mL of 0.2 M HCl by 0.2 M NaOH iii. 150 mL of 0.2 M HOCI (K₁ = 3.5 x 10-8) by 0.2 M NaOH iv. 150 mL of 0.2 MHF (K₁ = 7.2 Xx 10-4) by 0.2 M NaOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a Rank the four titrations in order of increasing pH at the halfway point to equivalence lowest to h...View the full answer

Answered By

S Mwaura
A quality-driven writer with special technical skills and vast experience in various disciplines. A plagiarism-free paper and impeccable quality content are what I deliver. Timely delivery and originality are guaranteed. Kindly allow me to do any work for you and I guarantee you an A-worthy paper.
4.80+
27+ Reviews
73+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Consider the following four titrations: i. 100.0 mL of 0.10 M HCl titrated with 0.10 M NaOH ii. 100.0 mL of 0.10 M NaOH titrated with 0.10 M HCl iii. 100.0 mL of 0.10 M CH3NH2 titrated with 0.10 M...
-
Catherine (aged 42) and Johnson (aged 45) have been married for 12 years. Johnson is a project manager of an event company at a monthly salary of $55,000 with an additional one-month salary of...
-
Please write detailed roadmap/solution for all questions below. 1) An output of nmap search is shown below, a) Type the required terminal command and required parameters to obtain the shown output....
-
The following are the Ledger Balance (in thousands) extracted from the books of Vaishnavi Bank Ltd as on March 31, 2016. The bank's Profit and Loss Account for the year ended and Balance Sheet as at...
-
Paul and Julie Leonards two-story home in Pascagoula, Mississippi, is only twelve feet above sea level and less than two hundred yards from the Gulf of Mexico. In 1989, the Leonards bought a...
-
Belda Co. makes organic juice in two departments: cutting and blending. Direct materials are added at the beginning of each process, and conversion costs are added evenly throughout each process. The...
-
The traditional income statement for Reeves Company shows sales \($900,000\), cost of goods sold \($600,000\), and operating expenses \($200,000\). Assum- ing all costs and expenses are 70% variable...
-
You have been approached by one of your clients, Mr. Sidney Chow, for help in determining what will be the tax consequences if he sells all his assets in his corporation (at their fair market value)...
-
E8-21 (Algo) Preparing Production and Direct Materials Purchases Budgets [LO 8-3b] Galactic Inc. manufactures flying drone toys. Sales units for January, February, March, April and May were 620, 600,...
-
From the following account balances, prepare in proper form for June (a) An income statement, (b) A statement of owner?s equity, (c) A balance sheet for Freeman Realty. Cash $5,200 S. Freeman,...
-
To what reaction does the solubility product constant, Ksp, refer? Table 15.1 lists K sp values for several ionic solids. For any of these ionic compounds, you should be able to calculate the...
-
Calculate the value of the equilibrium constant for each of the following reactions in aqueous solution. a. HCHO + OHCHO + HO b. C,H,O, +H*
-
Listed below are excess-enthalpy data at 25°C for two series of equimolar binary liquid mixtures. Explain why the mixture containing benzene is the "outlier" in each series. Series Mixture H IJ...
-
Using the Bollinger Bands chart The dates you could have bought the stock at the lowest possible price. The dates you could have sold the stock at the highest possible price. The behavior of the...
-
A flock of geese is attempting to migrate due south, but the wind is blowing from the west at 4.7m/s. a) If the birds can fly at 7.9m/s relative to the air, what direction should they head?
-
A box of volume 216 m with a square bottom and no top is made of two different materials. The cost of the bottom is $40/m and the cost of the sides is $30/m. Find the dimensions of the box that...
-
How can evidence-based stress management programs be integrated into educational curricula, healthcare settings, and workplace wellness initiatives to promote early intervention, resilience building,...
-
What challenges and opportunities does neurodiversity present in workplace settings, and how can organizations create environments that are inclusive of individuals with different neurocognitive...
-
The Harvard School of Public Health, in its College Alcohol Study Survey, surveyed college students in about 200 colleges in 1993, 1997, 1999, and 2001. The survey asked students questions about...
-
An environmentalist wants to determine if the median amount of potassium (mg/L) in rainwater in Lincoln County, Nebraska, is different from that in the rainwater in Clarendon County, South Carolina....
-
The malonic ester synthesis cannot be used to make 2,2-dimethylhexanoic acid. Explain why not.
-
When a malonic ester synthesis is performed using excess base and 1,4-dibromobutane as the alkyl halide, an intramolecular reaction occurs, and the product contains a ring. Draw the product of this...
-
Starting with ethyl acetoacetate and using any other reagents of your choice, propose an efficient synthesis for each of the following compounds. (a) (b) (c) OH
-
Use the australian accounting standard. On 1 July 2022, Surf Ltd leases a semi-trailer truck from Superior Ltd. The truck will be used to move spare parts between Surf's various warehouses. The...
-
In addition to the Transaction/events that have occurred during the year, the CEO would like you to provide some accounting advice on some proposed future expenditure. Specifically, Surf Ltd is...
-
A rise with a sharp fall at the end of an utterance is found in: a. statements b. yes/no questions c. exclamations d. wh-questions e. incomplete thoughts

Study smarter with the SolutionInn App


