Hydrazine (N 2 H 4 ) is used as a fuel in liquid-fueled rockets. When hydrazine reacts
Question:
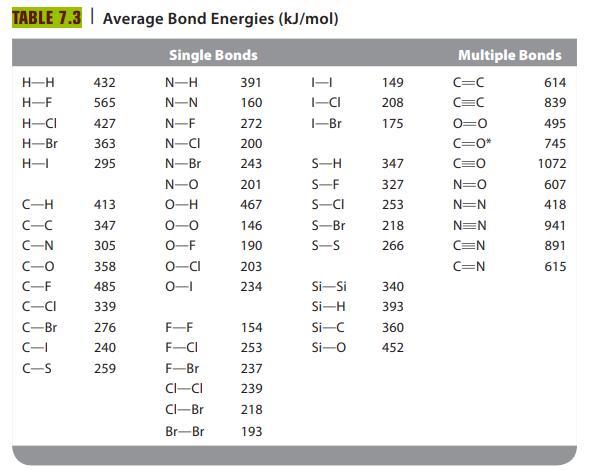
Hydrazine (N2H4) is used as a fuel in liquid-fueled rockets. When hydrazine reacts with oxygen gas, nitrogen gas and water vapor are produced. Write a balanced equation and use bond energies from Table 7.3 to estimate ΔH for this reaction.
Transcribed Image Text:
TABLE 7.3 Average Bond Energies (kJ/mol) Single Bonds N-H N-N H-H H-F H-CI H-Br H-1 C-H C-C C-N C-O C-F -CI C-Br C-I C-S 432 565 427 363 295 413 347 305 358 485 339 276 240 259 N-F N-CI N-Br N-O O-H 0-0 O-F O-CI 0-1 F-F F-CI F-Br CI-CI Cl-Br Br-Br 391 160 272 200 243 201 467 146 190 203 234 154 253 237 239 218 193 H I-CI 1-Br S-H S-F S-CI S-Bri S-S Si-Si Si-H Si-C Si-O 149 208 175 347 327 253 218 266 340 393 360 452 Multiple Bonds C=C C=C 0=0 C=O* C=0 N=O N=N N=N C=N C=N 614 839 495 745 1072 607 418 941 891 615
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The balanced chemical equation for the reaction between hydrazine N2H4 and oxygen ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Write an equation for the reaction of hydrazine with fluorine gas to produce nitrogen gas and hydrogen fluoride gas. Estimate ÎH for this reaction, using bond energies from Table 13.6. Table...
-
A promising new material with great potential as a fuel in solid rocket motors is ammonium dinitramide [NH4N(NO2)2]. a. Draw Lewis structures (including resonance forms) for the dinitramide ion...
-
The steering rockets in space vehicles use N 2 O 4 and a derivative of hydrazine, 1,1-dimethylhydrazine (Study Question 5.86). This mixture is called a hypergolic fuel because it ignites when the...
-
Problem 3.6.1 Given the random variable Y in Problem 3.4.1, let U = g(Y) = Y2. (a) Find Pu(u). (b) Find Fu(u). (c) Find E[U].
-
Powderhorn Corporation reported sales of $257,000, net income of $45,300, cash of $9,300, and net cash provided by operating activities of $21,200. Accounts receivable have increased at three times...
-
What are the advantages and disadvantages of e-mail surveys? What are situations when they may not be appropriate?
-
Enrique Inglesias, a sole trader, extracted the following trial balance from his books at the close of business on 31 July 2009: Required 1 Prepare the profit and loss accounts for the year ended 31...
-
Listed below are several terms and phrases associated with the FASBs conceptual framework. Pair each item from List A (by letter) with the item from List B that is most appropriately associated with...
-
D Question 11 Start Page 10 pts On September 1, Your Company issued 50,000 shares of $6 par value stock for $15 per share. Book value was $10 per share. Which journal entry records this transaction?...
-
Tom Belford and Tony Sorrentino own a small business devoted to kitchen and bath granite installations. Recently, building contractors have insisted on up-front bid prices for a house rather than the...
-
Although nitrogen trifluoride (NF 3 ) is a thermally stable compound, nitrogen triiodide (NI 3 ) is known to be a highly explosive material. NI 3 can be synthesized according to the equation a. What...
-
Give the Lewis structure, molecular structure, and hybridization of the oxygen atom for OF 2 . Would you expect OF 2 to be a strong oxidizing agent like O 2 F 2 discussed in Exercise 61? Exercise 61....
-
Matrix A represents a digital photograph. Find a matrix B that represents the negative image of 1. 030 131 2 32
-
A company developed the following per-unit standards for its product: 2 pounds of direct materials at $6.00 per pound. Last month, 3000 pounds of direct materials were purchased for $5700 and entered...
-
There are a number of ways that mental and emotional states can impact our health. Name 2 important aspects or dimensions of health psychology and describe how you might need improvement in those...
-
Budgeting is a tool used by management for performing the functions of planning, coordinating, and controlling operations of a business . Two main types of budgeting: static budgets and flexible...
-
2 -2 -41 Express the matrix A= -1 3 4 as the sum of a symmetric and skew -2-3] symmetric matrix. [10]
-
Consumer Corp. sells dishwashers and washing machines that come with a two - year unlimited warranty on parts and labour for repairs. The warranty is intended to assure customers that the appliances...
-
In Exercise 6.56, suppose that three of the customers are interested in the same car, and that they will go elsewhere if it has already been sold. Would it be appropriate to use the binomial...
-
Fill in each blank so that the resulting statement is true. A solution to a system of linear equations in two variables is an ordered pair that__________ .
-
VSEPR (valence state electron pair repulsion) theory was formulated to anticipate the local geometry about an atom in a molecule (see discussion in Section 25.1). All that is required is the number...
-
Chemists recognize that the cyclohexyl radical is likely to be more stable than the cyclopentylmethyl radical, because they know that six-membered rings are more stable than five-membered rings and,...
-
The presence of the carbonyl group in a molecule is easily confirmed by an intense line in the infrared spectrum around 1700 cm 1 that corresponds to a CO stretching vibration. Locate this line in...
-
Ida Company produces a handcrafted musical instrument called a gamelan that is similar to a xylophone. The gamelans are sold for $735. Selected data for the companys operations last year follow:...
-
Skull Company makes snowboards and uses the total cost method in setting product price. Its costs for producing 16,000 units follow. The company targets a 15.0% markup on total cost. Variable Costs...
-
Clue #6- Cash Disbursements for Selling and Administrative Expenses Monthly selling and administrative expenses are budgeted as follows: salaries and wages, $13,125 per month; shipping, 6% of sales;...

Study smarter with the SolutionInn App


