Kevlar, used in bulletproof vests, is made by the condensation copolymerization of the monomers Draw the structure
Question:
Kevlar, used in bulletproof vests, is made by the condensation copolymerization of the monomers 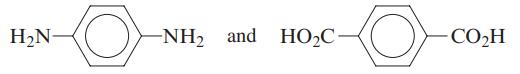
Draw the structure of a portion of the Kevlar chain.
Transcribed Image Text:
H₂N- O -NH₂ and HO₂C CO₂H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (5 reviews)
H2N benzene14diami...View the full answer

Answered By

Mary Njunu
I posses Vast, diversified knowledge and excellent grammar as a result of working in ACADEMIC WRITING for more than 5 years. I deliver work in various disciplines with assurance of quality work. I purpose at meeting the clients’ expectations precisely. Let’s work together for the best and phenomenal grades.
4.90+
928+ Reviews
2555+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Polyararrud is a term applied ro polyamides containing aromatic groups. These polymers were originally made for use as tire cords but have since found many other uses. a. Kevlar is used in...
-
Polyaramid is a term applied to polyamides containing aromatic groups. These polymers were originally made for use as tire cords but have since found many other uses. a. Kevlar is used in bulletproof...
-
Kevlar is a condensation polymer used in the manufacture of bulletproof vests. Identify the monomers required for the preparation of Kevlar. Kevlar
-
b) Calculate the Fault Level in MVA if a symmetrical bolted 3-phase fault occurs on the LV side of the 5MVA Transformer at 11kV c) The Tranformer impedance is 6% - Calculate the short circuit current.
-
Pierson Inc. operates a retail operation that purchases and sells snowmobiles, amongst other outdoor products. The company purchases all merchandise inventory on credit and uses a perpetual inventory...
-
Suppose that there are drastic technological improvements in shoe production at Home such that shoe factories can operate almost completely with computer-aided machines. Consider the following data...
-
Give examples of how a board member can increase the organization s social, intellectual, and fi nancial capacity.
-
Amy Austin established an insurance agency on March 1 of the current year and completed the following transactions during March: a. Opened a business bank account with a deposit of $50,000 in...
-
Problem 1 The air and walls are at 298K in a room where an insulated tube is going through the room. The outside diameter of the pipe is 0.07m, and its surface temperature and emissivity are 473K and...
-
Adam, Barbara and Charlotte formed the equal ABC partnership; Adam and Barbara each contributed cash of $100,000 and Charlotte contributed land worth $130,000 with a basis of $120,000 and subject to...
-
What kind of decay, if any, would you expect for the nuclei with the energy-level diagrams shown in Figure Q30.16? Energy (a) 22 22- Neutrons FIGURE Q30.16 Protons Energy (b) Neutrons 22 22- Protons...
-
CoCl 4 2- forms a tetrahedral complex ion and Co(CN) 6 3- forms an octahedral complex ion. What is wrong about the following statements concerning each complex ion and the d orbital splitting...
-
Two kg water at 120oC with a quality of 25% has its temperature raised 20oC in a constant volume process as in Fig. P5.45. What are the heat transfer and work in the process?
-
Find the electric field at the location of qa in the figure below, given that qb = c = ad = +2.30 nC, q = -1.00 nC, and the square is 15.0 cm on a side. (The +x axis is directed to the right.)...
-
8. Consider a perfectly conducting material, that is, a material for which the con- ductivity is infinite. Show that the magnetic field is time-independent inside a perfect conductor. Assume that in...
-
Identify and explain the difference(s) between horizontal and vertical audits. Explain the ways that customers and stakeholders might benefit from the changes that result from conducting an audit....
-
A company sells a single product with a contribution margin of $23 per unit. The company currently sells 1700 units per year and management believes that if they reduce the sales price by $1 per...
-
Content marketing is one of the key factors of new-age E-marketing. Elucidate on the benefits that content marketing helps you to achieve as a marketer? What are the major classifications of content...
-
1. To help the manufacturer get a clear picture of type I and type II error probabilities, draw a versus chart for sample sizes of 30, 40, 60, and 80. If is to be at most 1% with = 5%, which...
-
Let (x) = x 2 - 9, g(x) = 2x, and h(x) = x - 3. Find each of the following. (((--) 2
-
Use the geometrical construction shown in Example Problem 24.8 to derive the energy levels of the cycloheptatrienyl cation. What is the total energy of the molecule? How many unpaired electrons will...
-
One of the low-energy geometries of digermane, Ge 2 H 2 , is ethene-like. The Lewis dot structure shown
-
S p hybridization on each Ge atom in planar trans-digermane has been described as sp 1.5 for the GeGe sigma bond and sp 1.8 for the GeH bond. Suppose that the Ge lone electron (in terms of Lewis dot...
-
Assume there are three companies that in the past year paid exactly the same annual dividend of $2.73 a share. In addition, the future annual rate of growth in dividends for each of the three...
-
ces Assume that at the beginning of the year, you purchase an Investment for $5,160 that pays $106 annual income. Also assume the investment's value has decreased to $4,760 by the end of the year. a....
-
The bike you have been saving for is discounted 2 5 % . You have $ 6 0 0 saved to purchase it . The original, non - discounted price of the bike is $ 6 7 5 . There is a 5 . 7 7 % sales tax added to...

Study smarter with the SolutionInn App


