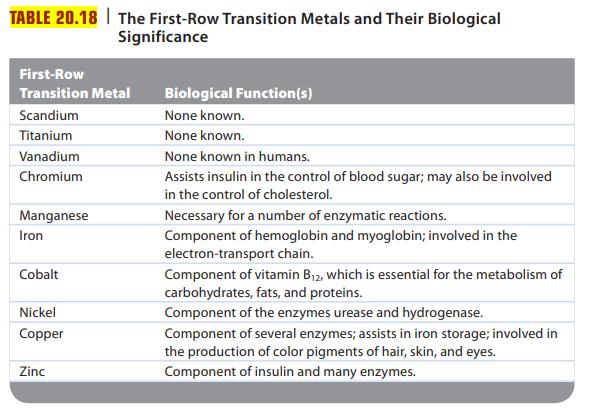
Review Table 20.18, which lists some important biological functions associated with different first-row transition metals. The transport
Question:
Review Table 20.18, which lists some important biological functions associated with different first-row transition metals. The transport of O2 in the blood is carried out by hemoglobin. Briefly explain how hemoglobin transports O2 in the blood.
Transcribed Image Text:
TABLE 20.18 The First-Row Transition Metals and Their Biological Significance First-Row Transition Metal Scandium Titanium Vanadium Chromium Manganese Iron Cobalt Nickel Copper Zinc Biological Function(s) None known. None known. None known in humans. Assists insulin in the control of blood sugar; may also be involved in the control of cholesterol. Necessary for a number of enzymatic reactions. Component of hemoglobin and myoglobin; involved in the electron-transport chain. Component of vitamin B₁2, which is essential for the metabolism of carbohydrates, fats, and proteins. Component of the enzymes urease and hydrogenase. Component of several enzymes; assists in iron storage; involved in the production of color pigments of hair, skin, and eyes. Component of insulin and many enzymes.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Hemoglobin is a protein found in red blood cells that transports oxygen from the lungs to the tissue...View the full answer

Answered By

Ankit Mahajan
I am an electrical engineering graduate from Thapar institute of engineering and technology.
Qualified exams - GATE 2019,2020.
CAT EXAM 2021- 91.4 percentile
SSC EXAMS- 2019,2020,2021
AFCAT EXAM- 2019,2020,2021
I want to share my knowledge with other people so that they can achieve the same.
I have strong hold Mathematics, Electrical engineering and all the subjects related.
Just give me a problem and I will give you the solution of it.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
The transport of O 2 in the blood is carried out by hemoglobin. Carbon monoxide (CO) can interfere with O 2 transport because hemoglobin has a stronger affinity for CO than for O 2 . If CO is...
-
Global Touch Corporation (GTC) is one of Canada's largest public companies. GTC provides end users with networking capabilities through its system of copper and coaxial cable lines. GTC operates in...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Explain why entrepreneurial firms are often in a strong position to use combination strategies.
-
An inexperienced accountant prepared this condensed income statement for Hight Company, a retail firm that has been in business for a number of years. HIGHT COMPANY Income Statement For the Year...
-
What are the managerial motivations that prove a source of conflict with marketing research?
-
Even though a businesss trial balance balanced, the following errors have been detected. You are required to prepare the correcting journal entries. a. A payment of \($550\) for rent was incorrectly...
-
Braverman Company has two manufacturing departments-Finishing and Fabrication. The predetermined overhead rates in Finishing and Fabrication are $18.00 per direct labor-hour and 110% of direct...
-
An article examined the graduate admissions process for evidence of gender bias. The table shows the number of applicants accepted to each of four graduate programs. % (Round to one decimal place as...
-
E3.6 Find Vo in Fig. E3.6 using nodal analysis.
-
Define and give an example of each of the following. a. Roasting b. Smelting c. Flotation d. Leaching e. Gangue What are the advantages and disadvantages of hydrometallurgy? Describe the process by...
-
Why do tetrahedral complex ions have a different crystal field diagram than octahedral complex ions? What is the tetrahedral crystal field diagram? Why are virtually all tetrahedral complex ions high...
-
State antidiscrimination statutes sometimes will expand protected class membership beyond the protections of federal law. a. True b. False
-
The case study is a Mass Casulty incident fire and the role is Patient Experience Manager at a Hospital Lessons Learned Report Directions. Your Report should address each Aspect requirement as...
-
7. Name two types of errors that might occur in the inventory account. If the error understates beginning inventory, how is cost of good sold impacted? If the error understates ending inventory, how...
-
3+x-5-x i) Prove that lim x-1 dy j) Find if x = at, y = 2at. dr
-
Should future Administrators seek FIO leadership with other backgroundsperhaps as insurance regulators in other countriesor is experience with insurance regulation at the state-level essential?
-
You observe the following preferred stock (a perpetuity) in the market: You can pay $1,000,000 today for the right to receive $70,000 annually starting in a year. What is the implied discount rate...
-
Unknown to a quality-control inspector, 20% of a very large shipment of electric switches are defective. The inspector has been told to reject the shipment if, in a sample of 15 switches, 2 or more...
-
For the data in Exercise 17-19, use the FIFO method to summarize total costs to account for, and assign these costs to units completed and transferred out, and to units in ending work in process....
-
Photoionization of a diatomic molecule produces a singly charged cation. For the molecules listed here, calculate the bond order of the neutral molecule and the lowest energy cation. For which of the...
-
What would the intensity versus frequency plot in Figure 25.10 look like if fluorescence were fast with respect to internal conversion? Fluorescence Absorption 0-4 Frequency So Absorption...
-
What aspect of the confocal microscope makes single-molecule spectroscopy in solutions possible?
-
For the fiscal year ended December 31, 2015, Brace Incorporated reported: Net income of $6,000, which includes Depreciation expense totaling $1,200 A loss on sale of equipment of $200 The company did...
-
Comparative financial statements for Weller Corporation, a merchandising company, for the year ending December 31 appear below. The company did not issue any new common stock during the year. A total...
-
Question 7 of 7 -/1 E Tom Zopf owns and manages a computer repair service, which had the following trial balance on December 31, 2021 (the end of its fiscal year). Concord Corporation Trial Balance...

Study smarter with the SolutionInn App


