The Hg 2+ ion forms complex ions with I - as follows: A solution is prepared by
Question:
The Hg2+ ion forms complex ions with I- as follows:
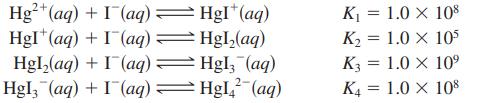
A solution is prepared by dissolving 0.088 mole of Hg(NO3)2 and 5.00 moles of NaI in enough water to make 1.0 L of solution.
![a. Calculate the equilibrium concentration of [HgI4]. b. Calculate the equilibrium concentration of [I]. c.](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1699/9/6/4/82265536796b3c181699964823549.jpg)
Transcribed Image Text:
Hg²+ (aq) + I (aq) = HgI+ (aq) + (aq) = (aq): Hgl₂(aq) + Hgl, (aq) + (aq) → = HgI*(aq) Hgl₂(aq) Hgl, (aq) HgI₂² (aq) K₁ = 1.0 X 108 K₂ = 1.0 X 105 K3 = 1.0 X 109 K4 1.0 X 108
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
The Hg2 ion forms complex ions with I as follows Hg2 I HgI HgI I HgI2 HgI2 I HgI3 HgI3 I HgI4 The eq...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
A solution is prepared by dissolving table salt, sodium chloride, in water at room temperature. a. Assuming there is no significant change in the volume of water during the preparation of the...
-
QUESTION 1 When propane undergoes complete combustion, the products are carbon dioxide and water.? ? ? ? __ C 3 H 8 (g) + __ O 2 (g) ? __ CO 2 (g) + __ H 2 O(g)What are the respective coefficients...
-
A solution is prepared by dissolving 10.8 g ammonium sulfate in enough water to make 100.0 mL of stock solution. A 10.00-mL sample of this stock solution is added to 50.00 mL of water. Calculate the...
-
Shown here is a list published by Electronics Weekly.com of the top five semiconductor companies in the United States by revenue ($ billions). a. Construct a bar chart to display these data. b....
-
Curtis is an executive on a business trip to the West Coast. He has driven his car on this trip and checks into the Hotel Ritz. The hotel has a guarded underground parking lot. Curtis gives his car...
-
TechPro offers instructional courses in e-commerce website design. The company holds classes in a building that it owns. Classify each of TechPros costs below as (a) Variable or fixed (b) Direct or...
-
You are contemplating buying stock in FMS Corp. FMS just paid a quarterly dividend of $1.00, and you expect future quarterly dividends to decline at a rate of 5 percent per quarter indefinitely. If...
-
A bag is gently pushed off the top of a wall at A and swings in a vertical plane at the end of a rope of length l Determine the angle for which the rope will break, knowing that it can withstand a...
-
E15-7 Admission of a Partner Pam and John are partners in PJ's partnership, having capital balances of $120,000 and $40,000, respectively, and share income in a ratio of 3:1. Gerry is to be admitted...
-
The following six-column table for Solutions Co. includes the unadjusted trial balance as of December 31, 2013. Required 1. Complete the six-column table by entering adjustments that reflect the...
-
Calculate the mass of manganese hydroxide present in 1300 mL of a saturated manganese hydroxide solution. For Mn(OH)2, K sp = 2.0 10 -13 .
-
Write equations for the stepwise formation of each of the following complex ions. a. Ni(CN) 4 2- b. V(C2O 4 ) 3 3 -
-
In Problems 944, find each indefinite integral and check the result by differentiating. ter xe dx
-
Marvel Parts, Incorporated, manufactures auto accessories. One of the company's products is a set of seat covers that can be adjusted to fit nearly any small car. The company has a standard cost...
-
A person has a near-point of 33.5 cm. They view an object through a microscope with a relaxed eye. The focal length of the objective lens is 1.5 cm and the distance between the two lenses if 15.8 cm....
-
Analyze the different conflict management styles used by employees. Include where each style might be appropriate through actual work examples.
-
Do research to identify the embedded operating systems used in BMW, Chevrolet, Chrysler, Ford, Honda, Mercedes Benz, Nissan, and Toyota automobiles. Use your creativity with this work. You are...
-
However, the skilled-based approach has potential challenges to use new skills effectively. Can you share more around that? I feel that both plans have benefits, but it depends on the organization,...
-
A study observes that the subjects in the study who say they exercise regularly reported only half as many serious illnesses per year, on the average, as those who say they do not exercise regularly....
-
What key concerns must functional tactics address in marketing? Finance? POM? Personnel?
-
Distinguish between the following terms applied to a set of functions: orthogonal and normalized, and orthonormal.
-
Why can we conclude that the wave function (x, t) = (x) e i (E / h )t represents a standing wave?
-
What is the usefulness of a complete set of functions?
-
Scenario You are an independent consultant hired by Penmen Groceries, and they are exploring options to modernize their accounting information system (AIS). They are looking for an audit company to...
-
Sort the following into asset, liability and expenses Accounts payable, Interest expense, Accounts receivable, Land, Advertising expense, Note payable, Building, Property tax expense, Cash, Rent...
-
The most commonly used style for a letter of application is Question 15 options: a) block letter style. b) adjusted block letter style. c) ragged right. d) ragged left

Study smarter with the SolutionInn App


