The formation of NO from N 2 and O 2 is unfavorable at 298 K, but it
Question:
The formation of NO from N2 and O2 is unfavorable at 298 K, but it becomes increasingly favored at high temperatures such as those in an automobile cylinder. Using data (ΔfH°, S° and ΔfG°) in Appendix L, calculate the equilibrium constant for the reaction ½ N2(g) + ½ O2(g) → NO(g) at 298 K and at 1000 K.
Data given in Appendix L
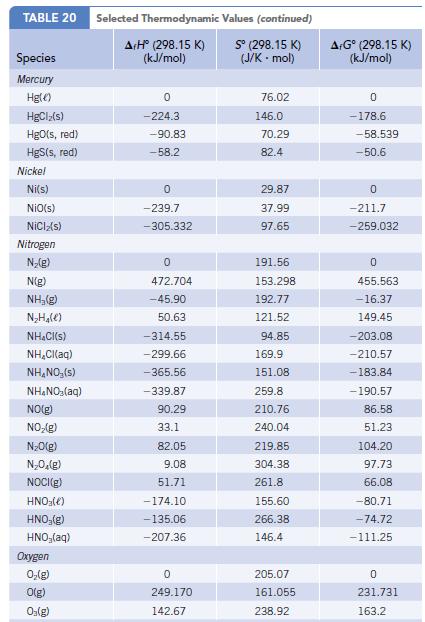
Transcribed Image Text:
TABLE 20 Species Mercury Hg() HgClz(s) HgO(s, red) HgS(s, red) Nickel Ni(s) NiO(s) NICIz(s) Nitrogen N₂(g) N(g) NH,(g) N₂H₂(e) NH₂Cl(s) NH₂Cl(aq) NH₂NO, (s) NH4NO₂(aq) NO(g) NO₂(g) N₂O(g) N₂O₂(g) NOCI(g) HNO3(e) HNO₂(g) HNO₂(aq) Oxygen 0₂(g) O(g) O₂(g) Selected Thermodynamic A+Hº (298.15 K) (kJ/mol) 0 -224.3 -90.83 -58.2 0 -239.7 -305.332 0 472.704 -45.90 50.63 -314.55 -299.66 -365.56 -339.87 90.29 33.1 82.05 9.08 51.71 -174.10 -135.06 -207.36 0 249.170 142.67 Values (continued) Sº (298.15 K) (J/K-mol) 76.02 146.0 70.29 82.4 29.87 37.99 97.65 191.56 153.298 192.77 121.52 94.85 169.9 151.08 259.8 210.76 240.04 219.85 304.38 261.8 155.60 266.38 146.4 205.07 161.055 238.92 A+Gº (298.15 K) (kJ/mol) 0 -178.6. -58.539 -50.6 0 -211.7 -259.032 0 455.563 -16.37 149.45 -203.08 -210.57 -183.84 -190.57 86.58 51.23 104.20 97.73 66.08 -80.71 -74.72 -111.25 0 231.731 163.2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Formation of NO from N2 and O2 The formation of NO from N2 and O2 is an endothermic reaction meaning ...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Explain, using Le Chtelier's principle, why the equilibrium constant for the formation of NO from N2 and O2 increases with increasing temperature, whereas the equilibrium constant for the formation...
-
The equilibrium constant (KP) for the formation of the air pollutant nitric oxide (NO) in an automobile engine at 530°C is 2.9 Ã 10-11: (a) Calculate the partial pressure of NO under these...
-
Write sentences for the long-term direction and strategic path that management intends to follow. "Where we are headed?" and should explain why the direction in which you intend to point the company...
-
Use the following information for this question: Taxable income Marginal tax rate 15% 25% 34% 39% 34% 35% S S 0-S 50,000 75,000 50,000-$ S 75,000 $100,000 $ 100,000-S 335,000 S 335,000-$10,000,000...
-
IBM recently saved $250 million over three years by implementing supply chain software that reduced the cost of components used in its manufacture of computers. If we assume that the savings occurred...
-
The following information was reported by three companies. When completing the requirements, assume that any and all purchases on account are for inventory. Required: 1. What amount did each company...
-
Which type of structure has decision making and power being led by a few people? A. Flat. B. Integrated. C. Matrix. D. Tall.
-
MagTech Inc. requires funding to build a new factory and has decided to raise the additional capital by issuing $850,000 face value of bonds with a coupon rate of 10%. In discussions with investment...
-
47 Please finish the sentence or answer the questions: 48 49 Observation on the maturity on the interest rate risk (read p.173 bottom and type in the two conclusions) As we will see momentarily, you...
-
Acetonitrile, CH 3 CN, is an important solvent. The chemical is normally available as a by-product of the manufacture of acrylonitrile, CH 2 =CHCN, the building block of polyacrylonitrile, a widely...
-
Chlorine atoms are formed by photochemical reactions of chlorofluorocarbons in the upper atmosphere. Using the average bond energy of the CCl bond in Table 8.8, calculate the wavelength of radiation...
-
Assume that the readings on the thermometers are normally distributed with a mean of 0C and a standard deviation of 1.00C. A thermometer is randomly selected and tested. In each case, draw a sketch,...
-
ABC Inc. has CAD20,000,000 interest payment due on September 19th and is concerned about a possible CAD appreciation. The premium for September 19th call option on Canadian dollar is $0.04, and the...
-
What does this code do? Explain what the following code snippet does in a couple of sentences. n = 1 while (n <1000): print(n) n = n * 5
-
Suppose the premium on a 9 1 - day 3 5 - strike XYZ Company stock call is $ 6 . 1 3 and the premium on a put with the same strike price is $ 0 . 4 4 , and the premium on a 9 1 - day 4 0 - strike XYZ...
-
Dividends on common stock during Year 2 ?totaled $ 1 5 ?thousand. The market price of common stock at the end of Year 2 ?was $ 1 6 . 3 0 ?per share. Required: Compute the following for Year 2 : ? a ....
-
The new president of the Blake Company was stumped. Why had profits gone down? He had directed the sales department to push the product with the highest contribution margin, and the sales department...
-
Wahoo, Inc., is a high-tech Internet company. It is trying to decide whether to issue NQOs or ISOs to its employees. Each employee will get 10 options. For purposes of this problem, assume that the...
-
Cobb Manufacturing Company uses a process cost system and average costing. The following production data is for the month of June 2011. Production Costs Work in process, beginning of the month:...
-
Consider the following circuit where the combinational circuit is represented by COMB and clock skew is represented by t skew . Given the following parameters: FF setup time = 20 ns FF hold time = 10...
-
Consider the following circuit where the combinational circuit is represented by COMB and clock skew is represented by tskew. Given the following parameters: FF setup time = 10 ns FF hold time = 2 ns...
-
A Mealy sequential machine has the following state table: Complete the following timing diagram. Clearly mark on the diagram the times at which you should read the values of Z. All state changes...
-
At 20 months, David loves playing with his blocks and has even attempted to build a 6-block tower, but his tower keeps falling down. His mother, Nicole, joins him when he plays with his blocks,...
-
A summary reconciliation of the government-wide and fund financial statements Must be presented at the bottom of the fund statements or in an accompanying schedule. Must be presented in the notes. Is...
-
Remember that statistics is basically counting things and making a scientifically supported decision about the results. Recently, many articles reported that the death rate for middle-aged Americans...

Study smarter with the SolutionInn App


