Use the standard reduction potentials (Appendix M) for the half-reactions Hg 2 Cl 2 (s) + 2e
Question:
Use the standard reduction potentials (Appendix M) for the half-reactions Hg2Cl2(s) + 2e− → 2 Hg(ℓ) + 2 Cl−(aq) and Hg22+(aq) + 2 e− → 2 Hg(ℓ) to calculate the value of Ksp for Hg2Cl2.
Data given in Appendix M
Transcribed Image Text:
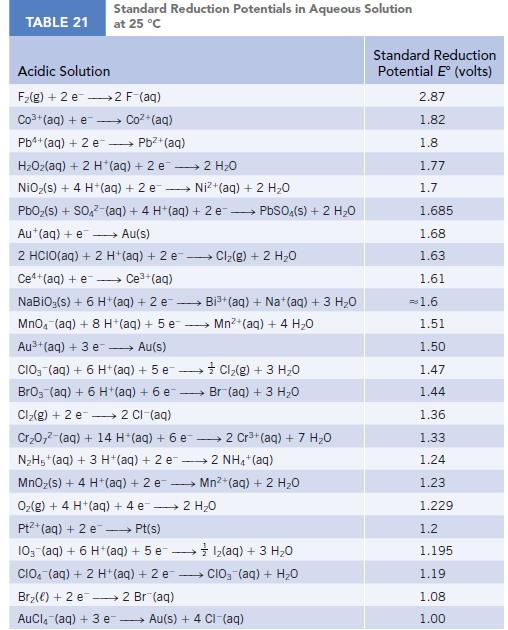
TABLE 21 Standard Reduction Potentials in Aqueous Solution at 25 °C Acidic Solution F₂(g) + 2 e 2 F-(aq) Co3+(aq) + e Coz+(aq) Pb4+ (aq) + 2 e - Pb²+ (aq) HzOz(aq) + 2 H*(aq) +2e → 2H2O NiO₂(s) + 4 H+ (aq) + 2 e→→→→→→ Ni+(aq) + 2 HO PbO₂ (s) + SO4² (aq) + 4 H+ (aq) + 2e → PbSO4(s) + 2 H₂O Au+ (aq) + e→→→→→ Au(s) 2 HCIO(aq) + 2 H+ (aq) + 2 e-- - Ce+(aq) + e→→→→ Ce³+ (aq) NaBiO;(s) + 6 H+ (aq) + 2 e- → MnO4 (aq) + 8 H+(aq) + 5 e Au³+ (aq) + 3 e→→→→→ Au(s) CIO3(aq) + 6 H+ (aq) + 5 e→→→→→→ BrO3 (aq) + 6 H+ (aq) + 6 e- Cl₂(g) + 2 e 2 Cl-(aq) Cr₂0,² (aq) + 14 H*(aq) + 6 e 2 Cr³+ (aq) + 7 H₂O N₂H5+ (aq) + 3 H+ (aq) + 2 e2 NH4+ (aq) MnO₂(s) + 4 H + (aq) + 2 e O₂(g) + 4 H+ (aq) + 4 e 2 H₂O Pt+ (aq) + 2 e →→→→→ Pt(s) 10- (aq) + 6 H+ (aq) + 5 e1₂(aq) + 3 H₂O CIO (aq) + 2 H+ (aq) + 2 e CIO₂ (aq) + H₂O Br₂() +2 e 2 Br(aq) AuCl(aq) + 3 e → Cl₂(g) + 2 H₂O → Bi³+ (aq) + Na+ (aq) + 3 H₂O Mn²+ (aq) + 4 H₂0 Cl₂(g) + 3 H₂O Br (aq) + 3 H₂O Mn²+ (aq) + 2 H₂O Au(s) + 4 CI-(aq) Standard Reduction Potential E (volts) 2.87 1.82 1.8 1.77 1.7 1.685 1.68 1.63 1.61 = 1.6 1.51 1.50 1.47 1.44 1.36 1.33 1.24 1.23 1.229 1.2 1.195 1.19 1.08 1.00
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 25% (4 reviews)
Hg2Cl2 2e 2 Hg 0 2 Claq Hg2aq 2e 2 Hg0 E 027 v E0789 v Ecell E oxidation ...View the full answer

Answered By

Sidharth Jain
My name is Sidharth. I completed engineering from National Institute of Technology Durgapur which is one of the top college in India. I am currently working as an Maths Faculty in one of the biggest IITJEE institute in India. Due to my passion in teaching and Maths, I came to this field. I've been teaching for almost 3 years.
Apart from it I also worked as an Expert Answerer on Chegg.com. I have many clients from USA to whom I teach online and help them in their assignments. I worked on many online classes on mymathlab and webassign. I guarantee for grade 'A'.
4.90+
3+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Determine the reactions at the beam supports for the given loading. 50 lb/in 400 lb 12 in 20 in 6 in
-
Use the standard reduction potentials (Appendix M) for the half-reactions [AuCl 4 ] (aq) + 3 e Au(s) + 4 Cl (aq) and Au 3+ (aq) + 3 e Au(s) to calculate the value of K formation for the complex...
-
Use the standard reduction potentials (Appendix M) for the half-reactions [Zn(OH) 4 ] 2 (aq) + 2 e Zn (s) + 4 OH (aq) and Zn 2+ (aq) + 2e Zn(s) to calculate the value of K formation for the...
-
Thor Bhd. (Thor) is a listed company in Malaysia, specializes in selling batteries. At 31 December 2021, Thor holds four distinct types of batteries in its warehouse. The accountant of Thor provided...
-
Determine the average rate of return for a project that is estimated to yield total income of $72,000 over three years, has a cost of $125,000, and has a $25,000 residual value.
-
Presented below is the ledger for Shumway Co. Instructions (a) Reproduce the journal entries for the transactions that occurred on October 1, 10, and 20, and provide explanations for each. (b)...
-
Equipment costing \($290,000,\) with an expected scrap value of \($30,000\) and an estimated useful life of five years, was purchased on January 1, 2016. Calculate the depreciation expense for years...
-
(Transactions, Financial StatementsService Company) Listed below are the transactions of Isao Aoki, D.D.S., for the month of September. Sept. 1 Isao Aoki begins practice as a dentist and invests...
-
3 20 points Princess Cruise Company (PCC) purchased a ship from Mitsubishi Heavy Industry for 580 million yen payable in one year. The current spot rate is 128 per dollar and the one-year forward...
-
In 1937, R. Schwartz and M. Schmiesser prepared a yellow-orange bromine oxide (BrO 2 ) by treating Br 2 with ozone in a fluorocarbon solvent. Many years later, J. Pascal found that, on heating, this...
-
The reaction occurring in the cell in which Al 2 O 3 and aluminum salts are electrolyzed is Al 3+ (aq) +3 e Al(s). If the electrolysis cell operates at 5.0 V and 1.0 10 5 A, what mass of aluminum...
-
For the following exercises, consider this scenario: A town has an initial population of 75,000. It grows at a constant rate of 2,500 per year for 5 years. Find a reasonable domain and range for the...
-
The importance and use of operations and quality management tools and how they contribute to achieving organizational objectives and competitiveness at PepsiCo ?
-
1. What is "interest rate risk"? 2. Which bond has higher interest rate risk,a long-term bondor a short-term bond, if all other features are the same? Why?
-
(a) Explain THREE (3) characteristics of Shariah compliant banking and financial system. (6 marks) (b) Explain sources of fund and uses of fund of Islamic banking and finance. (6 marks) (c) List FOUR...
-
A bank received $10,000.00 today as a final and unique payment for a loan that the borrower took 24 months ago at a 1.5% simple monthly rate. What was the principal of that loan? How much interest...
-
Joshua is traveling to Europe this summer. He wants to take the appropriate precautions to minimize any losses should there be any circumstances that would affect his trip. Joshua's mom has been ill,...
-
A currently profitable bricks-and-mortar retail firm is under attack from several Internet start-up firms. The top management has decided to join the Web competition and open up an Internet store....
-
A consumer magazine is evaluating five brands of trash compactors for their effectiveness in reducing the volume of typical household products that are discarded. In the experiment, each block...
-
This problem involves the design of a parallel adder-subtracter for 8-bit numbers expressed in sign and magnitude notation. The inputs X and Y are in sign and magnitude, and the output Z must be in...
-
Design a multiplier that will multiply two 16-bit signed binary integers to give a 32-bit product. Negative numbers should be represented in 2s complement form. Use the following method: First...
-
The objective of this problem is to use Verilog to describe and simulate a multiplier for signed binary numbers using Booths algorithm. Negative numbers should be represented by their 2s complement....
-
State the null and alternative hypothesis H0: Ha: 37 >37 >39 39 39
-
Content AreaHeedy Sparacia Winery accumulates the costs incurred in the labeling process in an activity cost pool. Costs for the labeling process are estimated to be $340,000, and the winery expects...
-
Explore the following through R-Commander in the selected datasets. a. Perform appropriate one-sample, two-sample mean, proportions, variance and ANOVA tests for your data and conclusions.All the...

Study smarter with the SolutionInn App


