Using data in Appendix L, calculate r G values for the decomposition of MCO 3 to
Question:
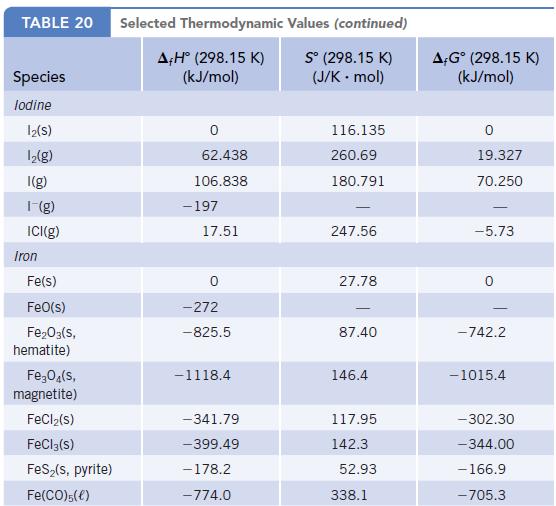
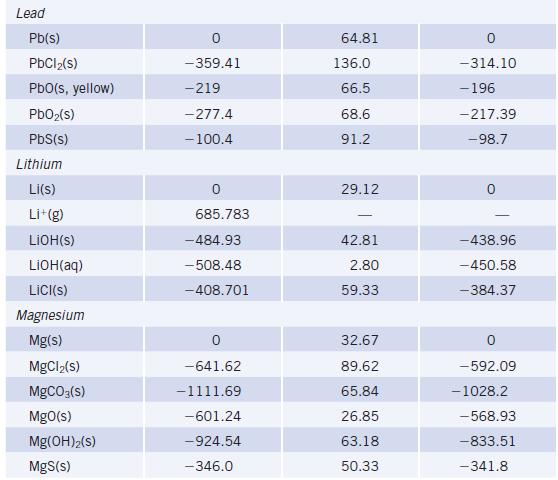
Using data in Appendix L, calculate ΔrG° values for the decomposition of MCO3 to MO and CO2 where M = Mg, Ca, Ba. What is the relative tendency of these carbonates to decompose?
Data given in Appendix L
Transcribed Image Text:
TABLE 20 Species lodine 12(S) 1₂(g) I(g) 1-(g) ICI(g) Iron Fe(s) FeO(s) Fe₂O3(s, hematite) Fe₂O4(s, magnetite) FeCl₂(s) FeCl3(s) FeS₂(s, pyrite) Fe(CO)5(€) Selected Thermodynamic A,Hº (298.15 K) (kJ/mol) 0 62.438 106.838 -197 17.51 -272 -825.5 -1118.4 -341.79 -399.49 -178.2 -774.0 Values (continued) Sº (298.15 K) (J/K.mol) 116.135 260.69 180.791 247.56 27.78 87.40 146.4 117.95 142.3 52.93 338.1 A-Gº (298.15 K) (kJ/mol) 19.327 70.250 -5.73 -742.2 -1015.4 -302.30 -344.00 - 166.9 -705.3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)

Answered By

Gilbert Chesire
I am a diligent writer who understands the writing conventions used in the industry and with the expertise to produce high quality papers at all times. I love to write plagiarism free work with which the grammar flows perfectly. I write both academics and articles with a lot of enthusiasm. I am always determined to put the interests of my customers before mine so as to build a cohesive environment where we can benefit from each other. I value all my clients and I pay them back by delivering the quality of work they yearn to get.
4.80+
14+ Reviews
49+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Using data in Appendix C, calculate ÎH°, ÎS°, and ÎG° at 298 K for each of the following reactions. In each case show that ÎG° = ÎH° -...
-
The decomposition of aqueous hydrogen peroxide in a given concentration of catalyst yielded the following data: Verify that the reaction is first order. Determine the rate constant for the...
-
Using data in Appendix E, calculate the standard emf for each of the following reactions: (a) H2(g) + F2(g) 2 H+ (aq) + 2 F- (aq) (b) Cu2+(aq) + Ca(s) Cu(s) + Ca2+ (aq) (c) 3 Fe2+ (aq) Fe(s) + 2...
-
A bond has an expected return of 15.33%, sensitivity to the term factor of 1.95 and to the default factor of 0.65. The current term risk premium is 3.01%. The risk-free rate of return is 3%. What is...
-
In the production-sales cycle, what are the four points at which quality costs are incurred? How are these costs interrelated through these points?
-
Crystal Charm Company makes handcrafted silver charms that attach to jewelry such as a necklace or bracelet. Each charm is adorned with two crystals of various colors. Standard costs follow: During...
-
Walgreens Boots Alliance, Inc. (Walgreens) is a global pharmacy, health and well-being company. It operates over 13,000 stores in 11 countries, generating net sales of \($103.4\) billion. The...
-
Georgia Woods, Inc., manufactures furniture to customers specifications and uses job order costing. A predetermined overhead rate is used in applying manufacturing overhead to individual jobs. In...
-
There were a lot of options given to me after entering my information, I used the smart planfinder to help narrow down the best choices. I compared the top three choices and they were all relatively...
-
Worldwide production of silicon carbide, SiC, is several hundred thousand tons annually. If you want to produce 1.0 10 5 metric tons of SiC, what mass (metric tons) of silicon sand (SiO 2 ) will you...
-
(a) Heating barium oxide in pure oxygen gives barium peroxide. Write a balanced equation for this reaction. (b) Barium peroxide is an excellent oxidizing agent. Write a balanced equation for the...
-
In Exercises 6368, find the solution set for each system by graphing both of the systems equations in the same rectangular coordinate system and finding points of intersection. Check all solutions in...
-
Banaag Manufacturing Company uses a job-order cost system in its two producing departments, Assembly and Finishing. Two jobs are in process at the beginning of the year and three more were started...
-
Convert the following code fragment: if (x == 1 || x == 2) switch (ch) { { case 'a': System.out.println("Good"); case 'b': System.out.println("Luck"); } } else if (x == 3 || x == 4) { if (ch 'c' ||...
-
Do companies incur any cost when they grant stock options to employees? If yes, who pays for these costs? What incentives do stock options create for the managers?
-
What are the driving forces of change in Mircosoft in which they competes? How has Mircosoft changed its strategy to address driving forces? Where does the company stand within the industry based on...
-
Time left 1:14:58 The market portfolio has an expected excess return of 6% and and a standard deviation of 15%. Asset A sits above the SML and has an expected excess return of 10% inclusive of alpha...
-
A newly established firm wants to establish a pension plan for its employees. The firm hires you to prepare a report comparing a defined benefit pension plan with a defined contribution pension plan....
-
For each of the following reactions, express the equilibrium constant: a) H20 (I) H2 (g) + 02 (g) Ke = 1.0x107 b) Fe2 (g) 2F (g) Ke= 4.9 x 10-21 c) C (s) + O2 (g) d) H2 (g) + C2H4 (g) C2H6 (g) Ke =...
-
In Section 5.4, we showed that the radius of a geosynchronous orbit about the Earth is 4.2 107 m, compared with the radius of the Earth, which is 6.4 106 m. By what factor is the force of gravity...
-
You are an astronaut (m = 95 kg) and travel to a planet that is the same radius and mass as the Earth, but it has a rotational period of only 2 h. What is your apparent weight at the equator of this...
-
Suppose the density of the Earth was somehow reduced from its actual value to 1000 kg/m 3 (the density of water). Find the value of g, the acceleration due to gravity, on this new planet. Assume the...
-
Your company will generate $68,000 in annual revenue at the end of each year for the next seven years from a new assembly line. If the appropriate interest rate is 8.5% p.a., compounded semi-...
-
Suppose that based on a survey conducted in BC and Ontario, 61% in BC and 54% in Ontario support the idea of carbon pricing. Suppose also that the sample sizes in BC and Ontario are 550 and 407,...
-
Seventy percent of children ages 12 to17 keep at least part of their savings in a savings account. You randomly select 48 children (ages 12 to 17) and ask them if they keep at least part of their...

Study smarter with the SolutionInn App


