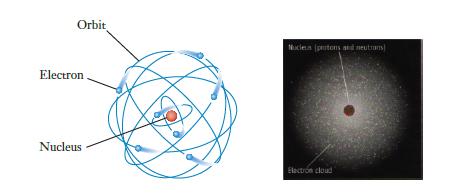
In a typical illustration of the atom such as Figure 2.3, which features lead to misunderstandings about
Question:
In a typical illustration of the atom such as Figure 2.3, which features lead to misunderstandings about the structure of atoms? Which ones give important insight?
Figure 2.3
Transcribed Image Text:
Orbit Electron Nucleus Nocleta (ptons and neutrons) Electron cloud
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
The image provided appears to show two models of the atom On the left there is a depiction of the Bohr model of the atom which presents electrons as p...View the full answer

Answered By

Deborah Joseph
My experience has a tutor has helped me with learning and relearning. You learn everyday actually and there are changes that are made to the curriculum every time so being a tutor has helped in keeping me updated about the present curriculum and all.
I have also been able to help over 100 students achieve better grades particularly in the categories of Math and Biology both in their internal examinations and external examinations.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
What is the effect of cultural commoditization and transformation on local traditions and customs? Give an example. Check Chapter 4 in the textbook for information on cultural commoditization and...
-
THIRD AVENUE SOFTWARE HEALTH-CARE APP PROJECT This case is new for the ninth edition of Information Technology Project Management . The case provides an opportunity to apply agile and Scrum...
-
Gothic Kings Ltd. Is a 100% owned subsidiary of Hadrian Inc. Gothic has been profitable in the past but incurred a loss for the year ended December 31, 20X3. Hadrian has indicated that if Gothic...
-
Refer to the financial statements and related notes of the Coca-Cola Company in Appendix A of this book. Required 1. Which depreciation method does the company use? Why do you think the company...
-
EMI Inc. is a public company that operates numerous movie theatres in Canada. Historically, it operated as a trust and its business model consisted of distributing all of its earnings to shareholders...
-
Use an activity-based costing system in a job-order environment.
-
Complete the table below for contribution margin per unit, total contribution margin, and contribution margin ratio: Number of units Sales price per unit Variable costs per unit Calculate:...
-
#11-A circle has a center at [-4,-1) and a point on the circle is located at (2.5). Write the standard form equation that would graph this circle.
-
Why is the number of protons called the atomic number?
-
One application of conductive polymers is in photovoltaic solar cells. Such devices have traditionally been silicon based. What possible advantages might conduct polymers offer?
-
A freight train rolls along a track with considerable momentum. If it rolls at the same speed but has twice as much mass, its momentum is (a) zero. (b) doubled. (c) quadrupled. (d) unchanged.
-
Frank Choy is normally a quiet person, but his patience has already been worn thin by interdepartmental battles. Choy joined Resonus Corporation, a hearing aid designer and manufacturer, eight months...
-
1. A circuit has three resistors connected to a 3.00-V battery in series: R = 15.02, R2 10.02, and R3 = 25.02. Find the equivalent resistance, Req, the total current in the circuit, Itot, and the...
-
all Q. The following data give the gain in body (kg) per heifer of two different breeds under a grazing treatment: Gain in body weight (kg) Breed Bread 51-3 26.9 53.2 16.8 44.8 54.2 71.4 Breed...
-
Q-2 Two samples drawn from two normal populations N (MI, a) and N The following information (42,922). 733 available on these samples regarding the expen diture in rupees per month per family. Sample...
-
Q.3 A sample of 400 families in an old city is selected ran randomly, and a sample of 500 families is randomly selected from several new colonies of the same city. A survey is conducted for the...
-
How important is the assumption "the sampled population is normally distributed" for the use of the chi-square distributions? Use a computer and the two sets of MINITAB commands that can be found in...
-
Why are stocks usually more risky than bonds?
-
Using acetylene and ethylene oxide as your only sources of carbon atoms, propose a synthesis for each of the following compounds. a. b.
-
Fill in the missing reagents below. Me OEt ?| Br Me SH OEt OMe OEt CN
-
Fill in the missing products below. excess HI Heat 1) Hg(OAc), Et 2) NABH, -: Na MCPBA 1) NaSH 2) H20 HBr
-
Telford Co. presented net income of $ 240,000, preferred dividends of $ 60,000 and common stock of 90,000 shares. The company also had $ 100,000 of convertible bonds. The bonds paid interest of $...
-
Wendell's Hot Sauce uses a process costing system to determine its product's cost. The last of the three processes is packaging. The Packaging Department reported the following information for the...
-
Consider the following data for two products of Gitano Manufacturing. (Round your intermediate calculations and OH rate and cost per unit answers to 2 decimal places) Number of units produced Direct...

Study smarter with the SolutionInn App


