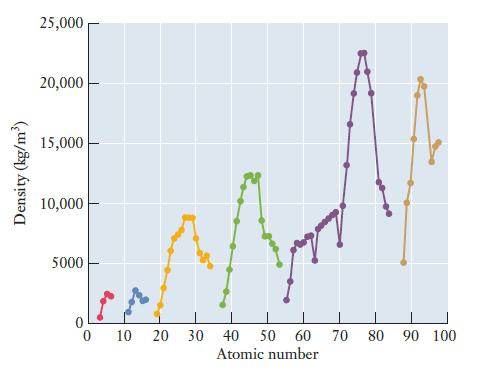
Most polymers have densities slightly less than 1 g/cm 3 . How does this compare to the
Question:
Most polymers have densities slightly less than 1 g/cm3. How does this compare to the densities of metals as shown in Figure 2.11? What does this comparison imply for devices in which conducting polymers might replace metal wires or other components?
Figure 2.11
Transcribed Image Text:
Density (kg/m³) 25,000 20,000 15,000 10,000 5000 0 10 20 30 40 50 60 70 80 90 100 Atomic number
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
To compare the densities we should first convert the given value to the units used in the ...View the full answer

Answered By

Felix Mucee
I am a detailed and thorough professional writer with 5 years of administrative experience- the last 2 years in academic writing and virtual office environment. I specialize in delivering quality services with respect to strict deadlines and high expectations. I am equipped with a dedicated home office complete with a computer, copier/scanner/fax and color printer.
I provide creative and detailed administrative, web search, academic writing, data entry, Personal assistant, Content writing, Translation, Academic writing, editing and proofreading services. I excel at working under tight deadlines with strict expectations. I possess the self-discipline and time management skills necessary to have served as an academic writer for the past five years. I can bring value to your business and help solve your administrative assistant issues.
4.70+
13+ Reviews
33+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
In our development of consumer theory, we made a big point about the fact that neoclassical economics does not put much stock in the idea of cardinally measuring utility (in terms of units of...
-
The investment banker helping Hologen with the Cybertech acquisition had done some preliminary research and concluded that Hologen could raise $200 million dollars by issuing a 5% coupon bond (paid...
-
The Silicon Valley company Intoot produces checkwriting software. The program itself, Fasten, sells for $50 and includes a package of checks. Check refill packets for Fasten cost $20 to produce and...
-
Consider the following set of data where an experiment was done to determine the molarity of a solute. What mass of sample is needed to reduce the %RSD to 0.10% if the sampling constant is 17.3 g?...
-
Prepare a short report that evaluates each of the following statements separately: 1. Since our plant was shut down for part of the year, we will not depreciate it. Depreciating it for the full year...
-
Brent Hill Company (see BEI0-21) borrowed $1 million on March I on a five-year, 12% note to help finance the building construction. In addition, the company had outstanding all year a $2-million,...
-
Prepare summary journal entries for the typical transactions of a job-costing system.
-
The following account balances appear in the 2015 adjusted trial balance of Blue Devils Corporation: Cash, $5,000; Accounts Receivable, $9,000; Supplies, $19,000; Equipment, $120,000; Accumulated...
-
onvert the following to logarithm form: 42=16 elect one: a. log 4216 b. log 2 16 4 c. log 4 16 = 2 d. log 164 2
-
Determine the name of the following ionic compounds: (a) Fe 2 O 3 , (b) Na 2 O, (c) Ca (NO 3 ) 2 Strategy We must determine the names of the constituent ions first. The anions will provide a hint...
-
The fact that a polymers physical properties depend on its atomic composition is very important in making these materials so useful. Why do you think this would be so?
-
Two cars, one a compact with mass 1200 kg and 1he other a large gas-guzzler with mass 3000 kg, collide head-on at typical freeway speeds. (a) Which car has a greater magnitude of momentum change?...
-
EX02-ME3150-21-24-W123.11 1.- Determine the R-value and the heat flux of One-Hour Wood Wall Assembly Dricon shown when convection coefficient inside and outside are 15 and 30 Btu/h ft2 F respectively...
-
Solve the below Equations : a.) y"-y=0 ; y(t) = et ; y2(t) = cosht
-
Problem 3 Engine oil enters at temperature T-75C a D-10 mm and Z-5 m long tube whose surface is maintained at T=175C. (a) What flow rate must be supplied for the outlet temperature to be TMD=100C?...
-
Prob 4. A sketch of a sewing machine is shown in Figure P1.24. Draw a kinematic diagram of the mechanism. Specify the number of links and the number of joints and calculate the mobility for the...
-
Topics: CLO2 Q2. Given an inductive displacement sensor in Figure Q2. (a) By sketching a central flux path and all related variables on the body of the sensor, estimate the inductance of the sensor...
-
Use a computer or calculator to find the p-value for the following hypothesis test: Ho: s2 = 7 versus Ha: s2 = 7 if x2 = 6.87 for a sample of n = 15
-
Explain the regulation of the secretions of the small intestine.
-
Epoxides can be formed by treating α-haloketones with sodium borohydride. Propose a mechanism for formation of the following epoxide. NABH,
-
When methyloxirane is treated with HBr, the bromide ion attacks the less substituted position. However, when phenyloxirane is treated with HBr, the bromide ion attacks the more substituted position....
-
Draw a Lewis structure of a carbon atom that has one extra valence electron (and therefore bears a negative charge). Which second-row element does this carbon atom resemble in terms of the number of...
-
The market value of Yeates Corporation's common stock had become excessively high. The stock was currently selling for $340 per share. To reduce the market price of the common stock, Yeates declared...
-
! Required information Use the following information for the Quick Study below. (Algo) (11-14) [The following information applies to the questions displayed below.] Trey Monson starts a merchandising...
-
Wakefield Associates is a recently formed law partnership. Ellery Hanley, the managing partner of Wakefield Associates, has just finished a tense phone call with Martin Offiah, president of Widnes...

Study smarter with the SolutionInn App


