Which of the following molecular-scale diagrams best represents a pure compound? Explain your answer. 8 8 (P)
Question:
Which of the following molecular-scale diagrams best represents a pure compound? Explain your answer.
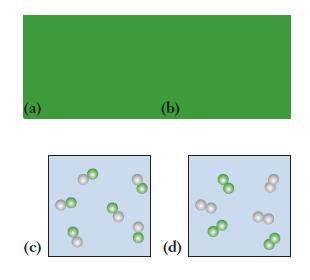
Transcribed Image Text:
€ 8 8 (P) со 800
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
The best molecularscale diagram that represents a pure compound is image b A pure comp...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
An average worker in Brazil can produce an ounce of soybeans in 20 minutes and an ounce of coffee in 60 minutes, while an average worker in Peru can produce an ounce of soybeans in 80 minutes and an...
-
Which of the following diagrams most likely represents an ionic compound, and which represents a molecular one? Explain your choice. (i) (ii)
-
Which of the following diagrams best represents a strong acid, such as HCl, dissolved in water? Which represents a weak acid? Which represents a very weak acid? (The hydrated proton is shown as a...
-
What are the roles and responsibilities of civil society in relation to the Sustainable Development Goals? In your country, how has civil society been engaged in the dialogue or discussion on...
-
During 2007, the controller of the Ryel Company asked you to prepare correcting journal entries for the following three situations: 1. Machine A was purchased for $50,000 on January 1, 2002....
-
What are the experimental units on which each of the following responses are observed? a. College GPA b. Household income c. Gasoline mileage rating for an automobile model d. Number of defective...
-
Net sales, net income, and total asset figures for Janice Controls, Inc., for five consecutive years are given below (Janice manufactures pollution controls): Required a. Calculate trend percentages,...
-
Better Days Ahead, a charitable organization, has a standing agreement with First National Bank. The agreement allows Better Days Ahead to overdraw its cash balance at the bank when donations are...
-
Association Between Smoking and Lung Cancer An epidemiological study was conducted to examine the relationship between smoking habits (current smoker vs. non-smoker) and the development of lung...
-
A student was given two metal cubes that looked similar. One was 1.05 cm on an edge and had a mass of 14.32 grams; the other was 2.66 cm on a side and had a mass of 215.3 grams. How can the student...
-
What type of transition is represented in the following molecular-scale illustration? co Coco co co COCO CO COCO CO CO co 88
-
Determine A in the indicated figures. Fig. 2.40 (c) B 4 66 A (c) 4 C
-
What is the most useful source of information on customers generated by any company? Identify all the possible data available on that source.
-
How does a data management system differ from a communication system?
-
List at least ten ambiguous words that should not be used in framing questions.
-
What are the criteria used for estimating the total effort required by a salesperson to cover a territory?
-
Who is currently in your own network that you could use for prospecting? How might you add to your network?
-
What is a disproportionation reaction? Use the following reduction potentials ClO3- + 3H+ + 2e- HClO2 + H2O o = 1.21 V HClO2 + 2H+ + 2e- HClO + H2O o = 1.65 V To predict whether HClO2 will...
-
Suppose Green Network Energy needs to raise money to finance its new manufacturing facility, but their CFO does not think the company is financially capable of making the periodic interest payments...
-
Determine the electron configuration for each of the following ions: a. A carbon atom with a negative charge b. A carbon atom with a positive charge c. A nitrogen atom with a positive charge d. An...
-
Identify which of the following two reactions you would expect to occur more rapidly: (1) addition of HBr to 2-methyl-2- pentene or (2) addition of HBr to 4-methyl-1-pentene. Explain your choice.
-
With current spectroscopic techniques (discussed in Chapters 1517), chemists are generally able to determine the structure of an unknown organic compound in just one day. These techniques have only...
-
(4 points) The annual claims made by clients on their insurance policies have an average of 10 million dollars and a standard deviation of 2 million dollars. If the amounts can be assumed to have a...
-
A researcher hypothesizes that inpatients who receive more visitors will self-report higher satisfaction with the level of care they receive. Which design would the researcher use to test the...
-
I like the answers in Ti -84 plus and using

Study smarter with the SolutionInn App


