The following boxes represent aqueous solutions containing a weak acid, HA and its conjugate base, A -
Question:
The following boxes represent aqueous solutions containing a weak acid, HA and its conjugate base, A-. Water molecules, hydronium ions, and cations are not shown. Which solution has the highest pH? Explain.
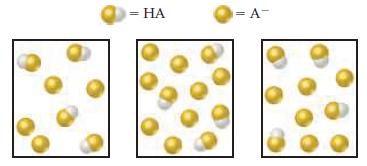
Transcribed Image Text:
- HA = A-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
Answer B The pH scale is a scale which measures how acidic ...View the full answer

Answered By

Enock Oduor
I am a chemist by profession, i coach high school students with their homework, i also do more research during my free time, i attend educational and science fair seminars where i meet students and do some projects.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry The Central Science
ISBN: 978-0134414232
14th Edition
Authors: Theodore Brown, H. LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward, Matthew Stoltzfus
Question Posted:
Students also viewed these Sciences questions
-
The following boxes represent aqueous solutions containing a weak acid, HX, and its conjugate base, X-. Water molecules, hydronium ions and cations are not shown. Which solution has the highest pH?...
-
A solution contains a weak monoprotic acid HA and its sodium salt NaA both at 0.1 M concentration. Show that [OH2] = Kw/Ka.
-
A weak acid HA (pKa 5.00) was titrated with 1.00 M KOH. The acid solution had a volume of 100.0 mL and a molarity of 0.100 M. Find the pH at the following volumes of base added and make a graph of pH...
-
According to adaptive expectations theory, a decline in the short-run aggregate demand curve ____ the inflation rate and ____ the unemployment rate. a) decreases; increases b) increases; decreases c)...
-
Mr. Eros operates an antique store located on the first floor of a four-story office building owned by Mr. Eros. The top three stories are leased to business tenants. Mr. Eros is considering giving a...
-
A case study in the chapter analyzed purchasing-power parity for several countries using the price of Big Macs. Here are data for a few more countries: a. For each country, compute the predicted...
-
The following summarised balance sheets relate to Track Limited: Additional information: 1. During the year to 30 June 2011, some fixed assets originally costing 25,000 had been sold for 20,000 in...
-
Linz Company sold $250,000 of 9.5 percent, 20-year bonds on April 1, 2014, at 106. The semiannual interest payment dates are March 31 and September 30. The market interest rate is 8.9 percent. The...
-
1) Overview of Note 5 in Apple's Financial Statements a) How much cash, cash equivalents, and marketable securities do Apple shareholders really own, net of the IRS claims on this cash? b) If Apple...
-
The monthly revenue R achieved by selling x wristwatches is figured to be R(x) = 75x 0.2x 2 .The monthly cost C of selling x wristwatches is C(x) = 32x + 1750. (a) How many wristwatches must the...
-
Predict the products of the following acidbase reactions, and predict whether the equilibrium lies to the left or to the right of the reaction arrow: (a) O 2- (aq) + H 2 O(l) (b) CH 3 COOH(aq) + HS...
-
Predict how each molecule or ion would act, in the Brnsted- Lowry sense, in aqueous solution by writing acid, base, both, or neither on the line provided. (a) HCO 3 - , the bicarbonate ion: ______...
-
A truck is traveling at 30 m/s on a slippery road. The driver slams on the brakes and the truck starts to skid. If the coefficient of kinetic friction between the tires and the road is 0.20, how far...
-
Write roles and responsibilities of Monitoring and Evaluation ( Youth development programe ) in Health Department?
-
Are all content types (text, visual, audio, and video) equally helpful for big companies vs. small businesses? Considering different social media platforms, how can different content be helpful for...
-
Relevance has always been the challenge of the advertising industry. Summarize Kristi Roger's assessment of the advertising industry in her TED Talk. Considering her hypothesis and results, what are...
-
Find the radius of convergence for the power series 2" (Inn)2z" and write down the open disk it is absolutely convergent.
-
After World War II , Japan's economy grew very fast. How would the Solow model explain this fact?
-
Green Leaf Fertilizer Ltd. experienced four situations for its supplies. Compute the amounts indicated by question marks for each situation. For situations 1 and 2, journalize the needed transaction....
-
You are planning to purchase your first home five years from today. The required down payment will be $50,000. You currently have $20,000. but you plan to contribute $500 each quarter to a special...
-
Sketch the shape and orientation of the following types of orbital: a. px, b. dz2 c. d x2 - y2
-
(a) What are the similarities and differences between the 1s and 2s orbital of the hydrogen atom? (b) In what sense does a 2p orbital have directional character? Compare the "directional"...
-
A certain quantum mechanical system has the energy levels shown in the diagram below. The energy levels are indexed by a single quantum number n that is an integer. (a) As drawn, which quantum...
-
Chapter 15 In-Class Learning Case Below is the Balance Sheet and Income Statement for Weaver Company as well as some additional information for the year ending on December 31, 2023. Balance Sheet...
-
A measurement that explains an accumulation of something over a given period of time is a O flow stock Ovalue asset
-
Answer is not complete. Increase/Decrease a. Correct change decrease b. Correct change decrease

Study smarter with the SolutionInn App


