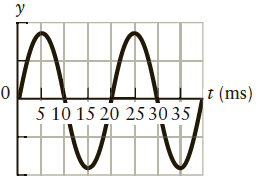
Consider a simple harmonic oscillator whose position as a function of time is given by Figure P11.38.
Question:
Consider a simple harmonic oscillator whose position as a function of time is given by Figure P11.38. At what times in this plot is the kinetic energy a maximum? At what time(s) is the potential energy a maximum?
Figure P11.38
?
Transcribed Image Text:
y t (ms) 5 10 15 20 25 30 35
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
Because the mass is constant the velocity determines the kinetic energy The velocity can be ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

College Physics Reasoning and Relationships
ISBN: 978-0840058195
2nd edition
Authors: Nicholas Giordano
Question Posted:
Students also viewed these Sciences questions
-
Consider a simple harmonic oscillator. Calculate the time averages of the kinetic and potential energies over one cycle, and show that these quantities are equal. Why is this a reasonable result?...
-
In describing the heat capacities of solids in Section 18.4, we stated that the potential energy U = 1/2kx2 of a harmonic oscillator averaged over one period of the motion is equal to the kinetic...
-
A simple harmonic oscillator consists of a 100-g mass attached to a spring whose force constant is 104 dyne/cm. The mass is displaced 3cm and released from rest. Calculate (a) The natural frequency...
-
The following data relating to direct materials cost for August of the current year are taken from the records of Happy Tots Inc., a manufacturer of plastic toys: Quantity of direct materials used...
-
Rather than purchase BLI directly (as in problems47 and 48), Amy and Brian will have their corporation, Spartan Tax Services (STS), acquire the business from Ernesto in a tax-deferred Type A merger....
-
A market researcher is interested in the average amount of money per year spent by students on entertainment. From 30 years of annual data, the following regression was estimated by least squares:...
-
Although not accompanied by an affidavit showing good cause, a subpoena that requests a nonparty to appear and testify at a deposition must be accompanied by witness fees and mileage costs, as set...
-
State Senator Bowdler convinced the legislature of State Z to pass a law requiring all professors to submit their class notes and transparencies to a board of censors to be sure that no lewd...
-
4. [-/1 Points] DETAILS MY NOTES OSCALC1 4.7.341. Draw the given optimization problem and solve. Find the volume of the largest right circular cylinder (in units) that fits in a sphere of radius 2...
-
According to a 2012 Business Travel News survey, the average salary of a travel manager is $110,550. Assume that the standard deviation of such salaries is $30,000. Consider a random sample of 50...
-
A particle attached to a spring with k = 50 N/m is undergoing simple harmonic motion, and its position is described by the equation x = (5.7 m)cos(7.5t), with t measured in seconds. (a) What is the...
-
The position of a simple harmonic oscillator is given by y = A sin(2pft), with f = 400 Hz. Find a value of t at which the potential energy is one quarter of its maximum value.
-
The following were selected from among the transactions completed by Essex Company during March of the current year: Mar. 2. Sold merchandise on account to Parsley Co., $32,000, terms 1/10, n/30. The...
-
Bramble Ltd., a retail store chain, had the following information in its general ledger for the year 2023: Merchandise purchased for resale $906,500 Interest on notes payable to vendors for the...
-
Case Scenario I Emma, a mid-level manager in a large manufacturing company, who wants to evaluate her personal performance in leading workplace relationships. Emma recognizes that effective...
-
What would the following code print? int[][] foo= new int[4][4]; for(int r = 0; r
-
Steam is admitted to the cylinder of an engine in such manner that the average pressure is 120 psi. The diameter of the piston is 10 inches and the length of stroke is 12 inches. How much work can be...
-
Joel de Paris, Incorporated Balance Sheet Beginning Balance Ending Balance Assets Cash $ 1 3 3 , 0 0 0 $ 1 2 7 , 0 0 0 Accounts receivable 3 4 2 , 0 0 0 4 7 5 , 0 0 0 Inventory 5 7 9 , 0 0 0 4 8 2 ,...
-
If a firm has no dividends and has negative earnings, which valuation models are appropriate?
-
Place a tick in the appropriate grid to identify the balance that would be brought down in each of the following named accounts, in the books of Rizwy Mohamed: (a) In the Cash account: if Rizwy...
-
Compute ave for H 2 O, HOD, and D 2 O. Do you need to perform the same calculation each time, or can you derive an expression that relates the ratio of average speeds for two gases to their...
-
Compare the average speed and average kinetic energy of O 2 with that of CCl 4 at 298 K. M o2 = 0.0320 kg mol -1 MccI 4 = 0.154 kg mol -1
-
How far on average does O 2 travel in 1 second at 298 K and 1 atm? How does this distance compare to that of Kr under identical conditions?
-
Consider a 1.00 m magnesium nitrate, Mg(NO3)2, aqueoussolution. a. Determine the molality (in m) of all solutes in the solution.Assume that magnesium nitrate completely ionizes when dissolved...
-
5. Cocoa powder, a main ingredient of chocolate, is made by roasting ground cacao beans (note the spelling difference). Caffeine and theobromine, two organic base compounds that produce the stimulant...
-
A body temperature of 97.0 F is slightly lower than the "normal" value but is still considered acceptable as an individual's temperature can have variable norms. What would this temperature equate to...

Study smarter with the SolutionInn App


