Using the Nyquist criterion, find the range of K for stability for each of the systems in
Question:
Using the Nyquist criterion, find the range of K for stability for each of the systems in Figure P10.4.
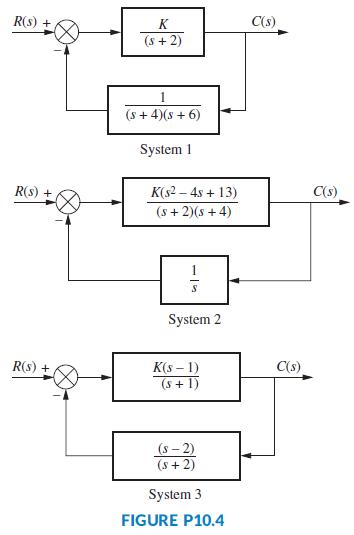
Transcribed Image Text:
R(s) + K C(s) (s + 2) (s + 4)(s + 6) System 1 R(s) + K(s2 – 4s + 13) C(s) (s + 2)(s + 4) System 2 K(s – 1) (s+ 1) R(s) + C(s) (s- 2) (s+ 2) System 3 FIGURE P10.4
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
The Nyquist criterion is a graphical method used to determine the stability of a control system b...View the full answer

Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Find the range of K for stability for the unity feedback system of Figure P6.3 with K(s + 4)(s 4) (s +3) G(s) R(s) + E(s) C(s) G(s) FIGURE P6.3
-
Sketch the root locus and find the range of K for stability for the unity feedback system shown in Figure P8.3 for the following conditions: a. b. K(s? + 1) G(s) (s - 1)(s+2)(s+ 3)
-
Use the Routh-Hurwitz criterion to find the range of K for which the system of Figure P6.6 is stable. R(s) + E(s) C(s) K(s? 2s + 2) 1 s2 + 2s +4 FIGURE P6.6
-
(7b-4c-1)-2 (2b3c-2)5 Simplify.
-
How do you think Tim and Chuck will respond to your response?
-
Blocker Company estimates ita uncollectible accounts based on an analysis of receivables. On December 31, a junior accountant prepared the following aging schedule for the company's $88,000 in...
-
__________ suggests that public corporations can function effectively even though their managers are self-interested and do not automatically bear the full consequences of their managerial actions....
-
Why is the liabilities section of the balance sheet of primary significance to bankers?
-
Quit smoking: In a survey of 444 HIV-positive than half of HIV-positive smokers have used a ni
-
In 2005, Bob Moyer was reviewing production costs for Mile High Cycles. Located in Denver, Colorado, the company sold very high-quality, handcrafted mountain bikes to bicycle retailers throughout the...
-
Use the results of Problem 17 to estimate the percent overshoot if the gain term in the numerator of the forward path of each part of the problem is respectively changed as follows: a. From 10 to 30...
-
Let in Figure P8.3. a. Plot the root locus. b. Write an expression for the closed-loop transfer function at the point where the three closed-loop poles meet. Ks+ 3 G(s) = (9 + s);s
-
The limiting current density for the reaction I3 + 2e 7 3 r at a platinum electrode is 28.9 A ern> when the concentration of KI is 6.6 X 10-4 mol dm3 and the temperature 25e. The diffusion...
-
degrees Given i(t)= 10cos (1000t+30) (A), and V30-93.63250.550. Find the phase of i in b 20 V20 www 10 10 20 d + 40 fi + 40mH V30 30 I
-
Munoz Company is considering investing in two new vans that are expected to generate combined cash inflows of $ 3 3 , 0 0 0 per year. The vans combined purchase price is $ 9 7 , 0 0 0 . The expected...
-
what distinguishes a chemical process from a physical process? give a chemical process that results to formation of salt when complete?
-
Use the following information for the Exercises below. ( Static ) BMX Company has one employee. FICA Social Security taxes are 6 . 2 % of the first $ 1 6 0 , 2 0 0 paid to its employee, and FICA...
-
A solution is made by dissolving 26.3 g of chromium(III)sulfate, Cr2(SO4)3, in enough water to make exactly 500 mL ofsolution. Calculate the concentration (molarity) of Cr2(SO4)3 inmol/L (M). M...
-
(a) Predict the mean value of y if x = 8. (b) Construct a 95% confidence interval for the mean value of y if x = 8. (c) Predict the value of y if x = 8. (d) Construct a 95% prediction interval for...
-
Sundial Technologies produces and sells customized network systems in New Brunswick. The company offers a 60-day, all software and labor-and an extra 90-day, parts-only- warranty on all of its...
-
A thermodynamicist claims to have developed a heat engine with 50 percent thermal efficiency when operating with thermal energy reservoirs at 1260 R and 510 R. Is this claim valid?
-
A heat engine is operating on a Carnot cycle and has a thermal efficiency of 75 percent. The waste heat from this engine is rejected to a nearby lake at 60F at a rate of 800 Btu/min. Determine (a)...
-
It is claimed that the efficiency of a completely reversible heat engine can be doubled by doubling the temperature of the energy source. Justify the validity of this claim.
-
(c) Using the information you obtained in part (b), check whether it is reasonable to assume that the two samples come from populations whose distributions have a common variance. (d) To investigate...
-
[STAT6032 ONLY] Use the Bonferroni method to compare the following treatments (i.e. these four specific level combinations of speed and depth): (2,3), (2,4), (3,3) 6%). Calculate the critical...
-
HW 1 Bevo T-shirt Company purchased land, paying $30,000 cash plus a $300,000 note payable. In addition, Bevo paid delinquent property tax of $2,000 and paid $14,000 to remove an unwanted building on...

Study smarter with the SolutionInn App


