The equilibrium constant for the conjugate acid-base pair is 8.00 x 10 -5 . From the additional
Question:
The equilibrium constant for the conjugate acid-base pair
![]()
is 8.00 x 10-5. From the additional information
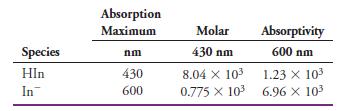
(a) Calculate the absorbance at 430 nm and 600 nm for the following indicator concentrations: 3.00 x 10-4 M, 2.00 x 10-4 M, 1.00 x 10-4 M, 0.500 x 10-4 M, and 0.250 x 10-4 M.
(b) Plot absorbance as a function of indicator concentration.
Transcribed Image Text:
HIn + H₂O = H₂0* + In¯
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
To calculate the absorbance at 430 nm and 600 nm for the different indicator concentrations we need ...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Analytical Chemistry
ISBN: 9780357450390
10th Edition
Authors: Douglas A. Skoog, Donald M. West, F. James Holler, Stanley R. Crouch
Question Posted:
Students also viewed these Sciences questions
-
The equilibrium constant for the following reaction is 1.0 Ã 10-3: Cr'. (aq) + H2EDTA2-(aq)--CrEDT A-(aq) + 2H' (aq) CH2-CO2 02GH CH EDTA N-CH,--CH2- O2C-CH2 CHy-CO2-...
-
At 2000 oC the equilibrium constant for the reaction Is Kc = 2.4 Ã 103. If the initial concentration of NO is 0.175 M, what are the equilibrium concentrations of NO, N2, and O2?
-
As shown in Table 15.2, the equilibrium constant for the reaction (a) What are the masses of N2 and H2 in the equilibrium mixture? (b) What was the initial mass of ammonia placed in the vessel? (c)...
-
Prove that the function x2 x - 1 | f (x) = x 1)(x 2) is differentiable for all r E (-0, 1) U (1, 2) U (2, ). - |
-
The reinvestment of capital gains and dividends can make a significant difference in your total return. Consider the following situation to determine the difference reinvestment can make over a...
-
Propylene is converted to butyraldehyde and n-butanol in the following reaction sequence: C3H6 + CO + H2 C3H7CHO (butyraldehyde) C3H7CHO + H2 C4H9OH (n-butanol) Liquid propylene, gaseous carbon...
-
The annual report of The Arcadia Company discloses the following amounts on its consolidated statement of income: Further, on Arcadias consolidated balance sheet, it reports the following amounts for...
-
Finer Company uses a sales journal, a purchases journal, a cash receipts journal, a cash disbursements journal, and a general journal. The following transactions occur in the month of May. May 2 Sold...
-
In the two-phase atomic commit protocol, each client has an uncertainty period after it has voted yes for a request (transaction) and is waiting for the final decision (commit or abort) from the...
-
Beale Management has a noncontributory, defined benefit pension plan. On December 31, 2021 (the end of Beale's fiscal year), the following pension-related data were available: Projected Benefit...
-
Three large proteins are ionized at the pH at which an electrical FFF separation is carried out. If the ions are designated A 2+ , B + , and C 3+ , predict the order of elution.
-
In Exercises find the limit graphically. Use the Sandwich Theorem to confirm your answer. lim x sin x
-
Dan Dayle started a business by issuing an $80,000 face-value note to First State Bank on January 1, Year 1. The note had an 8 percent annual rate of interest and a five-year term. Payments of...
-
Question 3 of 10 Wildhorse Enterprises provides the following information relative to its defined benefit pension plan. Balances or Values at December 31, 2025 Projected benefit obligation $2,727,900...
-
Delph Company uses job-order costing with a plantwide predetermined overhead rate based on machine-hours. At the beginning of the year, the company estimated that 50,000 machine-hours would be...
-
If a Hershey's Chocolate allows its lower level managers to aid in the decision making process because these managers are more aware of what is going on and need to develop their decision making...
-
Primare Corporation provided the following data for last month's manufacturing operations. Purchases of raw materials Indirect materials used in production Direct labor Manufacturing overhead applied...
-
Jarvene Corporation uses the FIFO method of process costing. The following data are for the most recent month of operations in one of the company's processing departments: Units in beginning...
-
Mason canceled a note issued by Emma (Mason's niece) that arose in connection with the sale of property. At the time of the cancellation, the note had a basis to Mason of $30,000, a face amount of...
-
Sue Deliveau opened a software consulting firm that immediately paid $2,000 for a computer. Was this event a transaction for the business?
-
Bromonium ions can be captured by nucleophiles other than water. Predict the products of each of the following reactions: a.
-
For each of the following objects determine whether or not it possesses a plane of symmetry: a. b. c. d. e. f.
-
Each of the following reactions proceeds via an S N 1 mechanism and will have anywhere from two to five steps, as discussed in Section 7.6. Determine the number of steps for each reaction, and then...
-
On Joe Martin's graduation from college, Joe's uncle promised him a gift of $11,100 in cash or $710 every quarter for the next 4 years after graduation. Assume money could be invested at 8%...
-
Retlaw Corporation ( RC ) manufactures time - series photographic equipment. It is currently at its target debt equity ratio of 0 . 7 3 . It s considering building a new $ 5 6 million manufacturing...
-
Elaborate on the use of sandbox while collaborating on wikis

Study smarter with the SolutionInn App


