Nitrogen at 600 kPa, 127C is in a 0.5-m 3 insulated tank connected to a pipe with
Question:
Nitrogen at 600 kPa, 127◦C is in a 0.5-m3 insulated tank connected to a pipe with a valve to a second insulated initially empty tank of volume 0.25 m3, shown in Fig. P6.158. The valve is opened, and the nitrogen fills both tanks at a uniform state. Find the final pressure and temperature and the entropy generation this process causes. Why is the process irreversible?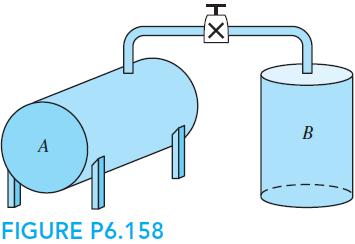
Transcribed Image Text:
В A FIGURE P6.158 HX
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
The final pressure and temperature of the nitrogen will be the same in both tanks after the process ...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
Nitrogen at 90 lbf/in .2 , 260Fis ina20 ft 3 insulated tank connected to pipe with a valve to a second insulated initially empty tank of volume 10 ft 3 . The valve is opened, and the nitrogen fills...
-
Saturated liquid nitrogen at 600 kPa enters a boiler at a rate of 0.005 kg/s and exits as saturated vapor. It then flows into a super heater, also at 600 kPa, where it exits at 600 kPa, 280 K. Assume...
-
Nitrogen at 600 kPa, 127C is in a 0.5 m3 insulated tank connected to a pipe with a valve to a second insulated initially empty tank of volume 0.5 m3. The valve is opened and the nitrogen...
-
Automobiles air bags are filled via the decomposition of sodium azide, according to the following equation: 2 NaN3 (s) 2 Na (s) +3 N2 (g) Calculate the work (in L atm) when 2.25 g of sodium azide...
-
Suppose that the U.S. is considering devaluing its dollar against a foreign currency to improve the trade balance. What type of pass-through effects would lead to a positive effect on the trade...
-
You are a practicing CPA at Gibbons, Johnson & Tannun, LLP. You recently received a new medium-sized client, ABI, Inc., a construction company that builds and renovates office buildings. Since the...
-
Gannon Company establishes a $400 petty cash fund on September 9. On September 30, the fund shows $166 in cash along with receipts for the following expenditures: transportation-in, $32; postage...
-
Circulating axial flow in an annulus (Fig. 3B.6), a rod of radius KR moves upward with a constant velocity v 0 through a cylindrical container of inner radius R containing a Newtonian liquid, the...
-
Peach Company makes 3 products in a single facility. The products have the following unit costs: Products A B C Direct Materials 12.50 14.40 10.30 Direct Labor 8.60 9.40 6.70 Variable MOH 1.10 2.20...
-
Required Information [The following information applies to the questions displayed below.] Ramer and Knox began a partnership by investing $54,000 and $81,000, respectively. The partners agreed to...
-
One kilogram carbon dioxide at 100 kPa, 400 K is mixed with 2 kg carbon dioxide at 200 kPa, 2000 K, in a rigid, insulated tank. Find the final state (P, T) and the entropy generation in the process...
-
Do the previous problem but assume that the process is isothermal. Data from previous problem A cylinder/piston contains 1 kg methane gas at 100 kPa, 300 K. The gas is compressed reversibly to a...
-
A water skier is towed 65 m to the south of the starting point, and then is turned 40 east of south for another 35 m. What is the skiers displacement from the starting point?
-
A company manufactures only custom furniture and uses a job order costing system to accumulate costs. Actual direct materials and direct labor costs are accumulated for each job, but a predetermined...
-
Obtain the following variables from Compustat North America's Fundamentals Annual data via WRDS: GVKEY (firm identifier), FYEAR, DLTT, DLC, CEQ, PRCC_F, CSHO, SICH, and SIC for all available...
-
Croy Incorporated has the following projected sales for the next five months: Month Sales in Units April 3 , 5 3 0 May 3 , 9 5 0 June 4 , 5 5 0 July 4 , 1 6 5 August 3 , 9 5 0 Croy s finished goods...
-
Question 1 You will be creating an invoicing system for the sales of PC parts. The file "Assignment_Q1.xlsm" contains the worksheets "Sales Point", "Items Database", and "Voucher Database". The...
-
JAVA LANGUAGE ONLY!!! I AM USING INTELLIJ IDEA COMMUNITY VERSION MAKE A MATRIX CALCULATOR INSTRUCTIONS ARE WRITTEN BELOW SEE 2ND PHOTO TO SEE WHAT SHOULD THE UML DIAGRAM OF THE CODE SHOULD LOOK LIKE...
-
You own a fixed-income asset with a duration of five years. If the level of interest rates, which is currently 8%, goes down by 10 basis points, how much do you expect the price of the asset to go up...
-
During 2012, Cheng Book Store paid $483,000 for land and built a store in Georgetown. Prior to construction, the city of Georgetown charged Cheng $1,300 for a building permit, which Cheng paid. Cheng...
-
A more accurate expression for E osc would be obtained by including additional terms in the TaylorMacLaurin series. The TaylorMacLaurin series expansion of f (x) in the vicinity of x 0 is given by...
-
The observed lines in the emission spectrum of atomic hydrogen are given by In the notation favored by spectroscopists, = 1/ = E/hc and R H =109,677 cm 1 . The Lyman, Balmer, and Paschen series...
-
Calculate the speed that a gas-phase fluorine molecule would have if it had the same energy as an infrared photon ( = 1.00 10 4 nm), a visible photon ( = 500. nm), an ultraviolet photon ( = 100....
-
On 1 January 20.1 SA Traders purchased office furniture for R80 000. The financial year-end of SA Traders is 31 December.Calculate the accumulated depreciation for 20.3 by using the reducing-balance...
-
Customs agents are on the lookout for situations where the value of the transaction (invoice value) may not represent the true value of goods being imported. Identify the 3 situations most likely to...
-
Iverson, Inc. has $1,294,000 of sales revenue, $692,000 of variable costs, and $692,000 of fixed costs in 2023. Assuming the contribution margin percentage stays the same in 2024, what will the break...

Study smarter with the SolutionInn App


