Redo the previous problem for a large stationary Brayton cycle where the low-T heat rejection is used
Question:
Redo the previous problem for a large stationary Brayton cycle where the low-T heat rejection is used in a process application and thus has nonzero exergy.
Data from previous problem
The conversion efficiency of the Brayton cycle in Eq. 10.1 was determined with cold air properties. Find a similar formula for the second-law efficiency, assuming the low-T heat rejection is assigned a zero exergy value.
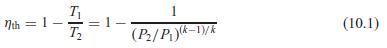
Transcribed Image Text:
T Nth = 1- T2 1 (10.1) (P/Pk-D/K
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
In order to redo the previous problem for a large stationary Brayton cycle with nonzero exergy for the lowT heat rejection we need to consider the exe...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
A large stationary Brayton cycle gas turbine power plant deliver output of 100 MW to an electric generator, minimum temperature in the cycle is 300 K and maximum temperature in the cycle is 1600 K,...
-
Find the power output and the low T heat rejection rate for a Carnot cycle heat engine that receives 6 kW at 250oC and rejects heat at 30oC as in Problem 7.35.
-
A large stationary Brayton cycle gas-turbine power plant delivers a power output of 100 MW to an electric generator. The minimum temperature in the cycle is 300 K, and the maximum temperature is 1600...
-
A portfolio manager owns a bond worth 2,000,000 that will mature in one year. The pound is currently worth $1.65, and the one-year future price is $1.61. If the value of the pound were to fall, the...
-
What are the IBFs? Why did the Federal Reserve authorize the establishment of the IBFs? Explain.
-
The budgets of four companies yield the following information: Company Requirements 1. Fill in the blanks for each company. 2. Compute breakeven, in sales dollars for each company. Which company has...
-
Xavier Construction negotiates a lump-sum purchase of several assets from a company that is going out of business. The purchase is completed on January 1, 2005, at a total cash price of $787,500 for...
-
Get It Right, CPAs, has been retained to review its client's corporate formation calculations for 20XX. Maria, Roger, and Novak created Grassroots Tennis, Inc. (GTI), which began operations on March...
-
Handy Leather, Inc., produces three sizes of sports gloves: small, medium, and large. A glove pattern is first stenciled onto leather in the Pattern Department. The stenciled patterns are then sent...
-
Lopez and Gomez are members in an LLC that reports net income of $130,000. An agreement states that Lopez receives a $20,000 salary allowance and Gomez receives a $50,000 salary allowance. Any...
-
A gasoline engine has a volumetric compression ratio of 9. The state before compression is 290 K, 90 kPa, and the peak cycle temperature is 1800 K. Find the pressure after expansion, the cycle net...
-
The conversion efficiency of the Brayton cycle in Eq. 10.1 was determined with cold air properties. Find a similar formula for the second-law efficiency, assuming the low-T heat rejection is assigned...
-
Fill in each blank with the correct response. For the arithmetic sequence having a n = 2n + 4, the term a 3 = _____.
-
A loan for $100000 is to be repaid with monthly payments for six years. The interest rate being charged is 3.4% compounded monthly. a. Find the size of the monthly payments. b. Find the final payment...
-
1. Choose a commercial service airport anywhere in the world and a public-use general aviation airport in the United States and prepare a one-page summary on each airport's (commercial and general...
-
Can you answer question one detailed and witihin this case thank you In 1989 Hoppe married a Danish lawyer named Catrine, whose parents owned a small restaurant in Copenhagen. When Catrine's...
-
Rick Rueta purchased a $91,000 home at 8% for 30 years with a down payment of $15,000. His annual real estate tax is $1,812 along with an annual insurance premium of $972. Rick's bank requires that...
-
a) Given is the program name OurCalculation with class Calculation that perform the addition, subtraction. Using the keywords EXTEND in inheritance, rewrite the code so that My Calculation will...
-
Using the historical risk premiums as your guide, what is your estimate of the expected annual HPR on the S&P 500 stock portfolio if the current risk-free interest rate is 5%?
-
Doorharmony Company makes doorbells. It has a weighted- average cost of capital of 5% and total assets of $ 5,900,000. Doorharmony has current liabilities of $ 750,000. Its operating income for the...
-
Identify the reagents necessary to produce each of the following compounds via an aldol reaction. (a) (b) (c) (d) (e)
-
Using formaldehyde and acetaldehyde as your only sources of carbon atoms, show how you could make each of the following compounds. You may find it helpful to review acetal formation. (a) (b) (c) (d)...
-
Draw a mechanism for the following transformation: NaOH, heat
-
The transactions in the journal and the accounts in the ledger are linked together by a transfer process called:Group of answer choicesaudit trailmigratingpostingbalancing
-
Garbo township recorded it's police departments appropriations for the year ending June 30, 2025. Police appropriations had increased to $7,600,000. During the year's first budgetary month ending...
-
QUALITY INDICATOR: Ninety-five percent of physician appointments/reappointments will be completed within 90 days of receipt of all required materials. medical staff office health information business...

Study smarter with the SolutionInn App


