Show that the van der Waals equation can be written as a cubic equation in the compressibility
Question:
Show that the van der Waals equation can be written as a cubic equation in the compressibility factor, as in Eq. 12.53.
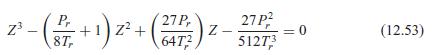
Transcribed Image Text:
2 () () - 27P, +1) z+ 6472 27P? 51273 z - = 0 (12.53)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
The van der Waals equation of state is given by P aV2 V b RT where P is the pressure V is the molar ...View the full answer

Answered By

User l_917591
As a Business Management graduate from Moi University, I had the opportunity to work as a tutor for undergraduate students in the same field. This experience allowed me to apply the theoretical knowledge I had gained in a practical setting, while also honing my teaching and communication skills.
As a tutor, I was responsible for conducting tutorial sessions, grading assignments and exams, and providing feedback and support to my students. I also assisted with the preparation of course materials and collaborated with other tutors and professors to ensure consistency in teaching and assessment.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
Show that van der Waals equation can be written as a cubic equation in the compressibility factor involving the reduced pressure and reduced temperature as (27 P 27 P2 512 T, (T+1) z2- - 1) z - Z3 ST...
-
Show that the van der Waals equation of state does not satisfy the criteria of intrinsic stability for all values of the parameters. Sketch the curves of P versus V for constant T (the isotherms of...
-
The van der Waals equation of state, an approximate representation of the behavior of gases at high pressure, is given by Where a and b are constants having different values for different gases. (In...
-
Parent Ltd owns 80% of Subsidiary Ltd. In the financial year ended 30 June 20X2, Subsidiary Ltd sold inventory to Parent Ltd. Details regarding the transaction are as follows: Cost to Subsidiary to...
-
What type of exchange rate system was the gold standard? Explain how it operated?
-
Sydney is interested in starting a new business, but would have to give up a job with a total compensation of $100,000 per year. After researching the new business opportunity, Sydney developed the...
-
The Metropolitan City Hospital is unionized. In 2006, nurses received an average annual salary of $45,000. The hospital administrator is considering how the contract with nurses should be changed for...
-
The 32 teams in the National Football League (NFL) are worth, on average, $1.17 billion, 5% more than last year. The following data show the annual revenue ($ millions) and the estimated team value...
-
Zoie makes 2 products from a common input. Each product may be sold at the split-off point or processed further. The following is known about each product: Product 1 Product 2 Allocated joint...
-
Client's Facts: The client is charged with burglary. He broke a window, entered his neighbor's garage, and took three cases of beer. The garage is a separate building located about six feet from the...
-
Show how to find the constants in Eq. 12.52 for the van der Waals EOS. Vc = 3b 27 R?T? a 64 P. (12.52) RT. b 8P.
-
Find the heat of evaporation, hfg, for R-134a at 0C from the generalized charts and compare to the value in Table B.5.
-
Figure 7.13 displays two groups of data points, given in Table 7.8. The convex hulls have also been plotted. It is possible to separate the two classes of points via a straight line. In fact, many...
-
Diego Company manufactures one product that is sold for $75 per unit in two geographic regions the East and West regions. The following information pertains to the company's first year of operations...
-
Kumar has been working for Dreamline Enterprises for 12 years. He has been a member of the company's defined benefit pension plan for the past ten years. Kumar plans to retire as of January 1st of...
-
Consider the following graph representing web page nodes and the edges (hyperlinks) connecting them. 1 5 3 4 2 6 1. Give the link probability matrix for the above graph. (10 points) 2. Let x be the...
-
Suburbia, Inc., sells one of its products for $150 each. Sales volume averages 800 units per year. Recently, its main competitor reduced the price of its product to $130. Suburbia expects sales to...
-
Mr Alexander paid a premium of $4 per share for one 3-month call option contract (one contract = 100 shares) of XYZ Corporation with a strike price of $55 per share. At the time of purchase, the XYZ...
-
When are expenses credited?
-
Inexhaustible collections of ONPOs are not required to be capitalized or depreciated, if certain criteria are met. Why is this so, and what accounting and reporting recognition, if any, is required...
-
For a pair of keto-enol tautomers, explain how IR spectroscopy might be used to identify whether the equilibrium favors the ketone or the enol.
-
Acrolein is an α,β-unsaturated aldehyde that is used in the production of a variety of polymers. Acrolein can be prepared by treating glycerol with an acid catalyst. Propose...
-
Draw the structure of the product with molecular formula C 10 H 10 O that is obtained when the compound below is heated with aqueous acid. CN CN C10H100 Heat
-
[STAT6032 ONLY] Use the Bonferroni method to compare the following treatments (i.e. these four specific level combinations of speed and depth): (2,3), (2,4), (3,3) 6%). Calculate the critical...
-
Provide a purchase journal entry of a product (50$ above), which is comprised of a rational cognition purchase decision, extensive problem-solving decision, ideal state opportunity recognition,...
-
12345 1 Income Statement 2021 2020 2019 2018 Period Ending: 30/06 30/06 30/06 30/06 5 Total Revenue 671.74 598.85 784.51 777.95 6 Revenue 671.74 598.85 784.51 777.95 7 Other Revenue, Total 8 Cost of...

Study smarter with the SolutionInn App


