Use Eq. 12.32 to solve for (T/P)s in terms of T, v, C p , and
Question:
Use Eq. 12.32 to solve for (∂T/∂P)s in terms of T, v, Cp, and αp. How large a temperature change does water at 25◦C (αp = 2.1 × 10−4 K−1) have when compressed from 100 kPa to 1000 kPa in an isentropic process?
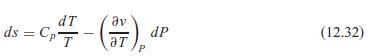
Transcribed Image Text:
dT ds = Cp av dP (12.32) aT
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 30% (13 reviews)
Starting with Eq 1232 ds CpT dT pV dP where s is entropy T is temperature P is pressure v is specifi...View the full answer

Answered By

Anik Kumar Bosu
I am expert in Maths, Physics, Chemistry and Biology. I also teach student from 3 years. Students becomes satisfied by my teaching. So, do not worry.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
2 m' of an ideal gas is compressed from 100 kPa to 200 kPa. As a result of the process, the internal energy of the gas increases by 10 kJ, and 140 kl of heat is lost to the surroundings. What is most...
-
Air is compressed from 100 kPa and 40C. The input power to the adiabatic compressor is 20 kW. Determine the mass flow rate of air through the compressor assuming the air undergoes a quasi-equilibrium...
-
Air is compressed from 100 kPa and 310 K to 1000 kPa in a two stage compressor with intercooling between stages. The intercooler pressure is 350 kPa. The air is cooled back to 310 K in the...
-
Cindy Bagnal, the manager of Cayce Printing Service, has provided you with the following aging schedule for Cayce's accounts receivable: Cindy indicates that the $126,700 of accounts receivable...
-
What is the difference between "dollarization" and a currency board?
-
Distinguish among the 5 design characteristics identified by Hackman and Oldham. Why is an understanding of these characteristics important for managers to know?
-
Denzel and Shantell form a partnership by contributing $70,000 and $35,000, respectively. They agree to an interest allowance equal to 10% of each partners capital balance at the beginning of the...
-
1. On what major assumptions is the argument by Marquis based? 2. What basic principles of abortion opponents and abortion rights supporters does Marquis cite? 3. How do advocates on both sides of...
-
PVC Inc. has a $15,000 loss from business operations this tax year, but the company did receive $90,000 in dividends from the 18%-owned BQE Inc., which is a domestic corporation. What is the total of...
-
A firm has $90,000 in direct materials costs and $60,000 in direct labor costs, with another $30,000 in overhead in 2022. It has equipment that was purchased for $900,000 and is being depreciated...
-
Use Eq. 12.34 to derive an expression for the derivative (T/v)s. What is the general shape of a constant s process curve in a Tv diagram? For an ideal gas, can you say a little more about the shape?...
-
Use Table B.3 to find the speed of sound for carbon dioxide at 2500 kPa near 100C. Approximate the partial derivative numerically.
-
On January 1, 2021, Sanderson Variety Store adopted the dollar-value LIFO retail inventory method. Accounting records provided the following information: Estimate ending inventory using the...
-
Itchen Magician, Inc. has assembled the following data pertaining to its two most popular products. Blender Electric Mixer Direct material$21$35 Direct labor1528 Manufacturing overhead @ $44 per...
-
Using R language 2) Write three ways to come up with all odd integers between 1 and 35 (including 1 and 35) 3) Using the ChickWeight dataset, provide a histogram of the weight variable using base R....
-
Based on our research, if we enter the market we would be competing in a competitive market. We have estimated the market demand and market supply for "mini" espresso machines to be Qd=9000-8P and...
-
Diego Company manufactures one product that is sold for $75 per unit in two geographic regions the East and West regions. The following information pertains to the company's first year of operations...
-
Diego Company manufactures one product that is sold for $75 per unit in two geographic regionsthe East and West regions. The following information pertains to the company's first year of operations...
-
Where should a business report cash which is restricted to purchase a long-term asset?
-
A firm offers two products for sale. The marginal cost of one product is new zero once the first unit has been produced. The marginal cost of the other product rises as output rises. What would be...
-
Draw a reasonable mechanism for the following transformation. NaOH, H,0 Heat
-
Predict the major product for each of the following transformations. (a) (b)
-
Propose an efficient synthesis for each of the following transformations. (a) (b) (c) (d) OEt EtO
-
$108,000 Current 162,000 Noncurrent On January 1, 2020, the balance sheet of Naperville Company (a sole proprietorship) was as follows. Assets Accounts receivable (net of allowance) Inventory...
-
Exercise 14-9 (Algo) Net Present Value Analysis and Simple Rate of Return [LO14-2, LO14-6] Derrick Iverson is a divisional manager for Holston Company. His annual pay raises are largely determined by...
-
Bramble Company has 1,920 pounds of raw materials in its December 31, 2021, ending inventory. Required production for January and February of 2022 are 3,840 and 4,800 units, respectively. Two pounds...

Study smarter with the SolutionInn App


