Verify that the ideal gas part of the Helmholtz function substituted in Eq. 12.86 does lead to
Question:
Verify that the ideal gas part of the Helmholtz function substituted in Eq. 12.86 does lead to the ideal gas law, as in the note after Eq. 12.97.
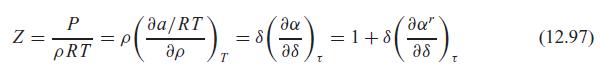
![]()
Transcribed Image Text:
da/RT da" = p pRT ap = 8 as =1+8 a8 (12.97) T
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
To verify that the ideal gas part of the Helmholtz function leads to the ideal gas law we can start ...View the full answer

Answered By

Joshua Marie Geuvara
I am an academic writer with over 5 years of experience. I write term papers, essays, dissertations, reports, and any other academic paper. My main objective is to produce a high-quality paper free from plagiarism and ensure a student scores an A+. Being a fluent English speaker, I have great communication skills that also enable me to produce excellent papers.
I am conversant with most academic referencing styles (APA, MLA, and Harvard).
You can trust me with your paper and expect nothing less than quality and excellent results. I look forward to meeting with you and, more importantly, developing something that will both make us happy and satisfied.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
Assuming that the ideal gas law holds, find the amount of nitrogen gas in a container if P = 0.836 atm 0.003 atm, V = 0.01985 m 3 0.00008 m 3 , T = 298.3 K 0.2 K. Find the expected error in the...
-
The ideal gas law relates four variables. An empirical gas law relates two variables, assuming the other two are constant. How many empirical gas laws can be obtained? Give statements of each.
-
The gas law for an ideal gas at absolute temperature T (in kelvins), pressure P (in atmospheres), and volume V (in liters) is PV = nRT, where is the number of moles of the gas and R = 0.0821 is the...
-
A machine fills containers with a particular product. The standard deviation of filling weights computed from past data is 0.6 ounces. If only 2% of the containers hold less than 18 ounces, what is...
-
Suppose 1 = $0.0077 in London, $1 = SF2.00 in New York, and SF1= 65 in Paris. a. If you begin by holding 10,000 yen, how could you make a profit from these exchange rates? b. Find the arbitrage...
-
Recent reports reveal market for RVs is faced with a many challenges over the next planning horizon. You are an analyst working at a consulting firm. Your firm has been hired by the auto industry to...
-
Part 1. Gibbs, Gier, and Gill are partners and share income and loss in a 5:1:4 ratio. The partner ships capital balances are as follows: Gibbs, $303,000; Gier, $74,000; and Gill, $223,000. Gibbs de...
-
Green Products Inc. cans peas and uses the weighted average cost method. For the month of November, the company showed the following: Peas completed and canned................. 245,000 lb Peas in...
-
Which of the following professional summaries is the strongest? Group of answer choices Hopeful college graduate seeing an a position in accounting. Detail-oriented recent accounting graduate. Strong...
-
Pop Corporation paid $686,000 for a 30 percent interest in Son Corporation's outstanding voting stock on April 1, 2016. At December 31, 2015, Son had net assets of $2,000,000 and only common stock...
-
R-410a is a 1:1 mass ratio mixture of R-32 and R-125. Find the specific volume at 20C, 1200 kPa, using Kays rule and the generalized charts, and compare it to the solution using Table B.4.
-
Gases like argon and neon have constant specific heats. Develop an expression for the ideal gas contribution to the Helmholtz function in Eq. 12.92 for these cases. a* = h* RT Ts* (12.92)
-
What does the size of the government spending multiplier relative to the size of the tax multiplier have to do with the effectiveness of fiscal policy at changing Real GDP?
-
How modern managerial accounting practices help companies grow Companies can find new ways to grow through forward-looking managerial accounting approaches. Improving business performance and...
-
What does Erica Olsen identify as the steps in the strategic planning process? How does the approach to strategic planning outlined by Erica Olsen in the video differ from the approach outlined in...
-
7. Stark Industries is a monopolistic competitor. It has fixed costs of $1,000 per month and a constant marginal cost of $1 per unit of production. (Hint: TC = FC + (MCQ)) a. If Stark Industries...
-
We had to read Leader-Member Exchange 'LMX' What Is It and Why You Should Care here:...
-
16 The total production cost for 20,000 units was P21,000 and the total production cost for making 50,000 units was P34,000. Once production exceeds 25,000 units, additional fixed costs of P4,000...
-
Where does the interest paid on bank loans get reported on the statement of cash flows?
-
Suppose the government bond described in problem 1 above is held for five years and then the savings institution acquiring the bond decides to sell it at a price of $940. Can you figure out the...
-
Rank the following compounds in terms of increasing basicity: N. Br z-
-
When (E)-4-amino-3-buten-2-one is treated with molecular hydrogen in the presence of platinum, the resulting amine is more basic than the reactant. Draw the reactant and the product, and explain why...
-
For each of the following compounds, draw the form that predominates at physiological pH: (a) (b) (c) CI CH CI Sertraline (Zoloft) An antidepressant NH2 Amantadine Used in the treatment of...
-
Since 1981, family-owned Southern Tool has grown into the ultimate destination for hard working do-it-yourselfers and professionals. Southern Tool Corporation has two divisions: Hand Tools and Power...
-
Sunland Inc. manufactures two electronic products, widgets and gadgets, and has a capacity of 1,700 machine hours. Prices and costs for each product are as follows: Widget Gadget Selling price per...
-
Spencer Company's inventory records for the most recent year contain the following data: (Click the icon to view the data.) Spencer Company sold a total of 19,200 units during the year. Read the...

Study smarter with the SolutionInn App


