Currently, CO 2 is being studied as a source of carbon atoms for synthesizing organic compounds. One
Question:
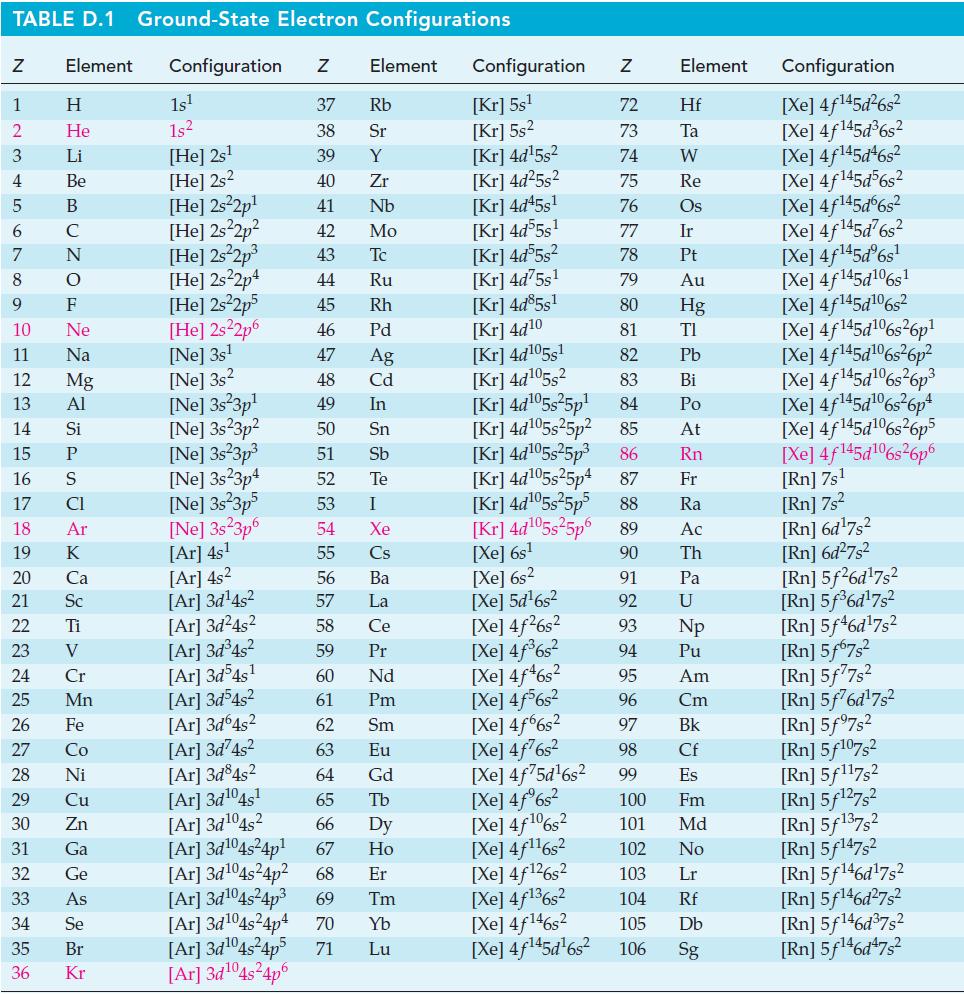
Currently, CO2 is being studied as a source of carbon atoms for synthesizing organic compounds. One possible reaction involves the conversion of CO2 to methanol, CH3OH.
![]()
With the aid of data from Appendix D, determine
(a) If this reaction proceeds to any significant extent at 25 °C;
(b) If the production of CH3OH(g) is favored by raising or lowering the temperature from 25 °C;
(c) K for this reaction at 500 K.
Transcribed Image Text:
CO₂(g) + 3 H2(g) CH3OH(g) + H₂O(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a To determine if the reaction CO2g 3 H2g CH3OHg H2Og proceeds to any significant extent at 25 C we can calculate the Gibbs free energy change of the ...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
The reaction between potassium superoxide, KO2, and CO2, 4 KO2 + 2 CO2 2K2CO3 + 3 O2 is used as a source of O2 and absorber of CO2 in self-contained breathing equipment used by rescue workers. (a)...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
Find the turning points on the curve y = 2x 3 5x 2 + 4x 1 and determine their nature. Find the point of inflection and sketch the graph of the curve.
-
LRF Companys budgeted sales and direct materials purchases are as follows. LRFs sales are 30% cash and 70% credit. Credit sales are collected 10% in the month of sale, 50% in the month following...
-
Sound propagating through air 30 oC at passes through a vertical cold front into air that is 4.0 oC. If the sound has a frequency of 2500 Hz, by what percentage does its wavelength change in crossing...
-
Information from a run ticket shows that 1,000 net barrels of oil with an API gravity of 36 were sold. The selling price is based on a contract price of $66/bbl, adjusted downward 4 for each degree...
-
On January 1, 2009, Plymouth Corporation acquired 80 percent of the outstanding voting stock of Sander Company in exchange for $1,200,000 cash. At that time, although Sanders book value was $925,000,...
-
55 2.5 What is the measure of arc KJ? 65 130 banana
-
To establish the law of conservation of mass, Lavoisier carefully studied the decomposition of mercury(II) oxide: (a) Show that the partial pressure of O 2 (g) in equilibrium with HgO(s) and Hg(l) at...
-
Two equations can be written for the dissolution of Mg(OH) 2 (s) in acidic solution. (a) Explain why these two equations have different r G values. (b) Will K for these two equations be the same or...
-
A loan company has been advertising on television a plan that allows people to borrow $1000 and make a payment of $10.87 per month. This payment is for interest only and includes no payment on the...
-
Answer these questions about Job Cost Accounting System What were the most compelling topics learned in this course? How did participating in discussions help your understanding of the subject...
-
A product costing $350, less 30%, 20%, and 10%, sells to allow for overhead expenses of 30% of the selling price and profit of 20% of the selling price. During a sale, the product is marked down by...
-
St Kilda Enterprises produces parts for electronics industry. The production manager and the cost analyst reviewed the accounts for previous accounts and have provided an estimated breakdown fixed...
-
CSC 226 Web Editor Component Lab You are part of a larger team that is building an HTML 5 editor. You have been given the task of creating dialogs for a set of web components allowing the web...
-
Caro Manufacturing has two production departments, Machining and Assembly, and two service departments, Maintenance and Cafeteria. Direct costs for each department and the proportion of service costs...
-
Explain why the electric field between two parallel plates of a capacitor decreases when a dielectric is inserted if the capacitor is not connected to a power sup-ply, but remains the same when it is...
-
Design an experiment to demonstrate that RNA transcripts are synthesized in the nucleus of eukaryotes and are subsequently transported to the cytoplasm.
-
Journal entries (continuation of 11-16). Refer to requirement 2 of Exercise 17-16. Prepare summary journal entries for the use of direct materials and incurrence of conversion costs. Also prepare a...
-
Zero beginning inventory, materials introduced in middle of process. Roary Chemicals has a Mixing Department and a Refining Department. Its process-costing system in the Mixing Department has two...
-
Weighted-average method, equivalent units. Consider the following data for the Assembly Division of Fenton Watches, Inc.: The Assembly Division uses the weighted-average method of process costing.
-
For the fiscal year ended December 31, 2015, Brace Incorporated reported: Net income of $6,000, which includes Depreciation expense totaling $1,200 A loss on sale of equipment of $200 The company did...
-
Comparative financial statements for Weller Corporation, a merchandising company, for the year ending December 31 appear below. The company did not issue any new common stock during the year. A total...
-
Question 7 of 7 -/1 E Tom Zopf owns and manages a computer repair service, which had the following trial balance on December 31, 2021 (the end of its fiscal year). Concord Corporation Trial Balance...

Study smarter with the SolutionInn App


