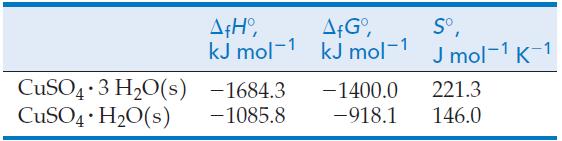
Use the following data together with other data from the text to determine the temperature at which
Question:
Use the following data together with other data from the text to determine the temperature at which the equilibrium pressure of water vapor above the two solids in the following reaction is 75 Torr.
![]()
Transcribed Image Text:
CuSO4 3 H₂O(s) — CuSO4 · H₂O(s) + 2 H₂0(g) . .
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
To determine the temperature at which the equilibrium pressure of water vapor above the two solids in the given reaction is 75 Torr we can use the Van...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Suppose that you have $250 to invest. a). You decide to store the $250 in cash under your mattress. If inflation is 3 percent, what will the real value of that cash be after one year under the...
-
In a study of the reaction 3Fe(s) + 4H2O(g) Fe3O4(s) + 4H2(g) at 1200 K, it was observed that when the equilibrium partial pressure of water vapor is 15.0 torr, the total pressure at equilibrium is...
-
Four vapor pressure data pointstwo representing solidvapor equilibrium and two representing liquidvapor equilibriumare available for a compound: A. Give your best estimate of the triple point...
-
Suppose you needed a material that could absorb heat without having its temperature increase very much. Would you choose aluminum or water? Make sure your explanation uses the concept of specific...
-
In Paige Company, direct labor is $20 per hour. The company expects to operate at 10,000 direct labor hours each month. In January 2014, direct labor totaling $204,000 is incurred in working 10,400...
-
An engineer redesigns a heat engine and improves its thermal efficiency from 20% to 25%. (a) Does the ratio of the heat input to heat output (1) increase, (2) remain the same, or (3) decrease?...
-
Study the format of the analysis of variances in Exhibit 12-12. Suppose production is 156,000 units. Also assume the following: Other data are as shown in Exhibit 12-12. Prepare an analysis of...
-
Herzogg Company, organized in 2010, has the following transactions related to intangible assets . InstructionsPrepare the necessary entries to record these intangibles. All costs incurred were for...
-
43 22 Cosine X 1 Which function would help you find x sine Tand
-
From the data given in Exercise 72, estimate a value of r S at 298 K for the reaction Exercise 72 Sodium carbonate, an important chemical used in the production of glass, is made from sodium...
-
The term thermodynamic stability refers to the sign of r G . If r G is negative, the compound is stable with respect to decomposition into its elements. Use the data in Appendix D to determine...
-
Complete the deposit ticket for Delectables Candies in Figure 4 - 18 The deposit is made on March 31, 20XX, and includes the following items: cash: $196.00; and check: Cavanaugh, $14,72; Bryan,...
-
4. What's wrong with the following instruction running on an ARM7TDMI? LDRSB r1, [r6], r3, LSL #4
-
magine you are in the process of planning to launch a new business venture in Canada. Your task is to conduct a comprehensive financial analysis to determine the initial funding requirements for your...
-
First, evaluate Gareth Hinds's use the graphic sound device (CLANG) in panel 15 as an interpretation of the text of the epic poem. Then, review lines 659-680 of the epic poem Beowulf , which describe...
-
# The Bells # Edgar Allen Poe theBells = ''' HEAR the sledges with the bells, Silver bells! What a world of merriment their melody foretells! How they tinkle, tinkle, tinkle, In the icy air of night!...
-
1 . Make a list of the industry's key success factors and measures of competitive strength or weakness. 2 . Assign weights to each competitive strength measure based on its perceived importance.
-
If it takes + 1.6 x 10-5J to move a positively charged particle between two charged parallel plates, (a) What is the charge on the particle if the plates are connected to a 6.0-V battery? (b) Was it...
-
The Cholesterol Level data sets give cholesterol levels of heart attack patients. Cholesterol measures are taken 2, 4, and 14 days aft er a patient has suffered a heart attack. Is there a significant...
-
Price and efficiency variances, journal entries. The Monroe Corporation manufactures lamps. It has set up the following standards per finished unit for direct materials and direct manufacturing...
-
Continuous improvement the Monroe Corporation sets monthly standard costs using a continuous-improvement approach. In January 2009, the standard direct material cost is $45 par unit and the standard...
-
Materials and manufacturing labor variances, standard costs. Dunn, Inc. is a privately held furniture manufacturer. For August 2009, Dunn had the following standards for one of its products, a wicker...
-
Larry's Woodworks has collected the following data for its cutting board line of products: Direct materials standard Direct materials standard cost Actual Direct Materials Used (AQU) Actual finished...
-
DIF MOH HCM 416 Use the information for St. Vincent's Hospital. Assume the hospital uses salary dollars as the costs driver for general administration and employee benefits, and space utilization as...
-
Create an Excel spreadsheet to solve the following problems. 1. Daniel deposited a lump sum 10 years ago, earned 3% interest compounded monthly and now has $40,000. How much did he invest? a. The NBC...

Study smarter with the SolutionInn App


