Use the information provided in Figure 21-5 to write balanced chemical equations for the reactions involved in
Question:
Use the information provided in Figure 21-5 to write balanced chemical equations for the reactions involved in synthesizing sodium carbonate from sodium chloride.
Figure 21-5
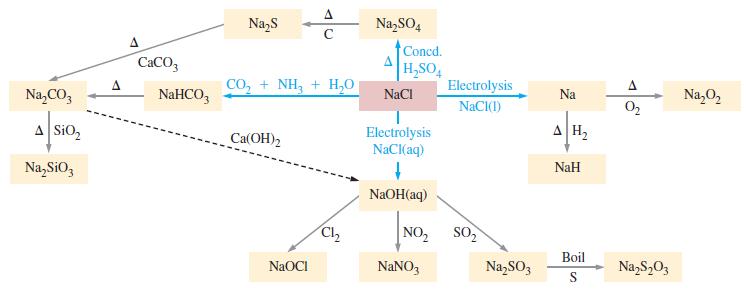
Transcribed Image Text:
Na₂CO3 A SiO₂ Na₂SiO3 A CaCO3 NaHCO3 Na₂S CO, + NH, + H,O Ca(OH) 2 A C NaOCI Cl₂2 Na₂SO4 Concd. H₂SO4 NaCl Electrolysis NaCl(aq) ↓ NaOH(aq) NO₂ NaNO3 Electrolysis NaCl(1) SO₂ Na₂SO3 Na AH₂ NaH Boil S A 0₂ Na₂S₂03 Na₂O₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
Analyze Consult Figure 215 to find a route from NaCl to Na 2 CO 3 One possib...View the full answer

Answered By

William Otieno
I am a professional tutor and a writer with excellent skills that are important in serving the bloggers and other specialties that requires a great writer. The important aspects of being the best are that I have served so many clients with excellence
With excellent skills, I have acquired very many recommendations which have made it possible for me to survive as an excellent and cherished writer. Being an excellent content writer am also a reputable IT writer with essential skills that can make one turn papers into excellent result.
4.70+
83+ Reviews
354+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Iron is extracted from its ores haematite and magnetite. Usually it is extracted from haematite (iron(iii) oxide). The ore is mixed with limestone and coke and reduced to the metal in a blast...
-
Copy and complete the table below, which shows the results of the electrolysis of four substances using inert electrodes. a. State what you understand by inert electrodes. b Explain why the lithium...
-
A 6.53-g sample of a mixture of magnesium carbonate and calcium carbonate is treated with excess hydrochloric acid. The resulting reaction produces 1.72 L of carbon dioxide gas 28oC at and 743 torr...
-
Each table of values gives several points that lie on a line.(a) What is the x-intercept of the line? The y-intercept?(b) Which equation in choices AD corresponds to the given table of values?(c)...
-
You have recently been hired as a staff assistant in the office of the chairman of the board of directors of Clefton, Inc. Because you have some background in accounting, the chairman has asked you...
-
Allan Stojanovich can purchase an office desk for $1,500 with cash terms of 2/10, n/30. If he can borrow the money at 12% annual simple ordinary interest for 20 days, will he save money by taking...
-
In ____________ communication the sender is likely to be most comfortable, whereas in ____________ communication the receiver is likely to feel most informed. (a) one-way; two-way (b) top-down;...
-
The steady-state distribution of temperature on a heated plate can be modeled by the Laplace equation, 0 = ? 2 T/?x 2 + ? 2 T/?y 2 If the plate is represented by a series of nodes (Figure), centered...
-
3. Professional Ethics: Analyze and discuss whether or not you think that the same ethical theory is being used at Zappos currently. Then considering the needs of stakeholders from a social and/or...
-
Use information from the chapter to write chemical equations to represent each of the following: (a) Reaction of cesium metal with chlorine gas; (b) Formation of sodium peroxide (Na 2 O 2 ); (c)...
-
Listed are several pairs of substances. For some pairs, one or both members of the pair react individually with water to produce a gas. For others, neither member of the pair reacts with water. The...
-
Can mental exercise build "mental muscle"? In one study of this question, 12 littermate pairs of young male rats were used; one member of each pair, chosen at random, was raised in an "enriched"...
-
Define marriage and critical rules associated with it in different cultures.
-
Anthropologists have long been interested in kinship, especially in societies without centralized government. Why should this be the case?
-
Evaluate the state of religion in U.S. society.
-
Summarize the characteristics of fundamentalism and discuss its relation to social change.
-
Analyze the differences among nuclear, composite, and extended families and the societies in which they're found.
-
College student Jacqueline Loya asked students who had full-time jobs and students who had part time jobs how many times they went out to eat in the last month. Briefly compare the distributions of...
-
With your classmates, form small teams of skunkworks. Your task is to identify an innovation that you think would benefit your school, college, or university, and to outline an action plan for...
-
Does the expected rate of return on the portfolio depend on the percentage of the portfolio invested in each stock? What about the riskiness of the portfolio?
-
Suppose an investor starts with a portfolio consisting of one randomly selected stock. What would happen (1) to the riskiness and to the expected return of the portfolio as more randomly selected...
-
Should the effects of a portfolio impact the way investors think about the riskiness of individual stocks?
-
q1: q2: help and explain The Museum of History is preparing for its annual appreciation dinner for contributing members. Last year, 650 members attended the dinner. Tickets for the dinner were $26...
-
This is part 2. The following information is from the Oregon Revised Statutes, Chapter 673, for Tax Consultants and Tax Preparers. Click the following link to access the reading material for part 2...
-
The manager explained that this practice created a pool of money that could be refunded to departments when they were having financial difficulties. It's also known that department managers receive a...

Study smarter with the SolutionInn App


