Indicate the molecular geometry around each carbon atom in the compound H;CCH=CHCH,CCH,COOH
Question:
Indicate the molecular geometry around each carbon atom in the compound
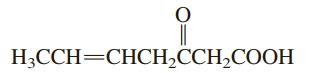
Transcribed Image Text:
H;CCH=CHCH,CCH,COOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
Numbering the seven carbon atoms from left to right the ...View the full answer

Answered By

HARSH RANJAN
Taken classes at college to graduates, Also worked as an expert to a freelancer online question-solving portal for more than 8 months with an average rating greater than 4.2 out of 5.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following samples has a larger mass? O 19.3 g of Au 8.92 10 atoms of Pt Identify the molecular geometry around the three selected atoms. Treat that atoms as the central atom for each...
-
Search Textbo a. chloroform, CHCI3 (carbon is central atom) Lewis structure Total number of electron groups around the central atom Electron geometry Molecular shape b. Carbon tetrachloride, CCI4...
-
The lactic acid molecule, CH3CH (OH) COOH, gives sour milk its unpleasant, sour taste. (a) Draw the Lewis structure for the molecule, assuming that carbon always forms four bonds in its stable...
-
What is the coefficient of sliding friction and what is a representative value for this coefficient for the brittle crust?
-
A coagulation-microfiltration process for removing bacteria from water was investigated in Environmental Science & Engineering (Sept. 1, 2000). Chemical engineers at Seoul National University...
-
Column (a) in Table 13.1 shows the monthly return on the British FTSE 100 index from August 1999 through July 2001. Columns (b) and (c) show the returns on the stocks of two firms. Both announced...
-
You are to enter up the sales, purchases and the returns inwards and returns outwards journals from the following details, then to post the items to the relevant accounts in the sales and purchase...
-
Baldwin Inc. is an athletic footware company that began operations on January 1, 2014. The following transactions relate to debt investments acquired by Baldwin Inc., which has a fiscal year ending...
-
hello, i need an answer about those questions and a deep explanation about it, thank you 1. On February 24, 2021, the Hong Kong stock market planned to raise the trading tax for the whole Hong Kong...
-
If the consumption function is given by C = 4200 + 0.75Y, state the marginal propensity to consume and deduce the marginal propensity to save.
-
Draw the cis and trans isomers of the compound 3-methyl-3-hexene.
-
Complete and balance the reaction for each of the following undergoing a combustion reaction. a. 2-methylpentane b. 2,2-dimethylpropane c. 3-methyl-3-hexene d. 3-ethyl-3-propanol e. ethylbenzene
-
Write notes on. (i). Transport-costing (ii). Power house costing (iii). Canteen costing.
-
Consider the regression \(Y_{i}=\beta_{0}+\beta_{1} X_{i}+u_{i}\), where \(Y_{i}\) denotes a worker's average hourly earnings (measured in dollars) and \(X_{i}\) is a binary (or indicator) variable...
-
Why are environmental problems often said to be simply a problem of common ownership?
-
Illustrate the effect of the destruction of the oil fields of Saudi Arabia on the oil market. Explain what then happens to the market for gasoline.
-
Give the linearization assumption and governing equation of slender axis-symmetric body.
-
Explain why the U.S. system of payment for medical procedures leads to higher health care costs than a system of payment for physicians services.
-
1. SUW, Inc uses standard costs to value its inventories. The standard cost of materials is $27 (9lbs @ $3 per lb). During the current month, 150,000 lbs were purchased at $2.95 per lb and 140,000...
-
Charles owns an office building and land that are used in his trade or business. The office building and land were acquired in 1978 for $800,000 and $100,000, respectively. During the current year,...
-
a. When 0.10 mol of the ionic solid NaX, where X is an unknown anion, is dissolved in enough water to make 1.0 L of Concept Explorations solution, the pH of the solution is 9.12. When 0.10 mol of the...
-
Which of the following beakers best represents a container of a weak acid, HA, in water? (Water molecules have been omitted for clarity.) ,- = A- = HA A
-
You have 0.10-mol samples of three acids identified simply as HX, HY, and HZ. For each acid, you make up 0.10 M solutions by adding sufficient water to each of the acid samples. When you measure the...
-
AC 0 ezto.mheducation.com 13e_ch15_final student - Fa23 SAMG 1200-22... G11. What is a 'proper promotional mix' - Google Sea... Chapter 14 Homework by Sunday, 11/12 i M Question 8-Chapter 14 Homework...
-
23 BUSM 1260-22 Applied Business Mathematics/Calculators ezto.mheducation.com M Question 1- Chapter 14 Homework by Sunday, 11/12 - Connect 4 Homework by Sunday, 11/12 ok nt rences Complete the...
-
ABC Bank has assets of $120 and a leverage ratio of 3. It earns 5% on its assets but pays 4% on its liabilities. What is ABC Bank's profit per unit of capital invested? X (round to the nearest...

Study smarter with the SolutionInn App


