For an electron in the hydrogen state find (r) after first expressing it in terms of a
Question:
For an electron in the hydrogen state
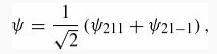
find (r) after first expressing it in terms of a single reduced matrix element.
Transcribed Image Text:
4 = 1 금 (V211 + 21-1),
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
The expectation value of r is and applying Eq...View the full answer

Answered By

Amar Kumar Behera
I am an expert in science and technology. I provide dedicated guidance and help in understanding key concepts in various fields such as mechanical engineering, industrial engineering, electronics, computer science, physics and maths. I will help you clarify your doubts and explain ideas and concepts that are otherwise difficult to follow. I also provide proof reading services. I hold a number of degrees in engineering from top 10 universities of the US and Europe.
My experience spans 20 years in academia and industry. I have worked for top blue chip companies.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introduction To Quantum Mechanics
ISBN: 9781107189638
3rd Edition
Authors: David J. Griffiths, Darrell F. Schroeter
Question Posted:
Students also viewed these Engineering questions
-
A hydrogen atom has an electron in the n = 5 level. (a) If the electron returns to the ground state by emitting radiation, what is the minimum number of photons that can be emitted? (b) What is the...
-
An excited hydrogen atom with an electron in the n = 5 state emits light having a frequency of 6.90 1014 s-1. Determine the principal quantum level for the final state in this electronic transition.
-
For an electron in the ground state of a hydrogen atom, calculate its (a) Potential energy, (b) Kinetic energy, and (c) Total energy.
-
Find reactions of support 1,2 and 3 F=10KN, a=2m, ?a=60 (45%) F 2a
-
A survey is taken to estimate the mean annual family income for families living in public housing in Chicago. For a random sample of 29 families, the annual incomes (in hundreds of dollars) are as...
-
How much energy is consumed by (a) A 1.2 kW hair dryer used for 10 min. (b) A 10 W night light left on for 24 h?
-
Which of the following requires UK limited companies to publish financial information? a. The Companies Act b. The Corporation Act c. European Union Directive 421B d. The Partnership Act
-
Code Churn is a common metric used to measure the efficiency and productivity of software engineers and computer programmers. It?s usually measured as the percentage of a programmer?s code that must...
-
Consider the following information pertaining to OldWest's inventory: Net Realizable Product Quantity Cost Value Revolvers 17 $129 $157 Spurs 27 25 20 Hats 9 57 47 At what amount should OldWest...
-
1. Take a position either for or against the following statement: To prepare for disasters, you should stockpile inventory, other critical supplies, and raw materials. 2. Think of Heinzs (H. J. Heinz...
-
Work out pH (t) for the system in Example 6.7 and comment on the correspondence with the classical equation of motion. Example 6.7 A particle of mass m moves in one dimension in a harmonic-oscillator...
-
Show that the commutator [L,f] = 0 leads to the same rule, Equation 6.46, as does the commutator [L,f] = 0. (n'lm| lm) = (n'l (m + 1) | (m +1)). (6.46)
-
Sara Bayer started a small merchandising business in Year 1. The business experienced the following events during its first year of operation. Assume that Bayer uses the perpetual inventory system....
-
Edwina bought a chargeable asset in August 2012 for 240,000, paying acquisition costs of 12,000. In June 2019 she sold a one-quarter interest in the asset for 100,000 and incurred disposal costs of...
-
David was given a chargeable asset in November 2017 at which time the asset had a market value of 7,500. He sold the asset in January 2024. Compute the chargeable gain or the allowable loss if his...
-
Prove that at the azeotropic point, the composition of the vapour and liquid phases are the same.
-
Discuss the significance of liquid-liquid equilibrium in extraction process.
-
What do you mean by depression of freezing point? Discuss van't Hoff's law of freezing point depression of a solution.
-
These transactions took place for Bramson Co. 2016 May 1 Received a $5,000, 12-month, 6% note in exchange for an outstanding account receivable from R. Stoney. Dec. 31 Accrued interest revenue on the...
-
Can partitioned join be used for r r.A s? Explain your answer
-
A foundation measuring 1.2 m 2.4 m in plan is constructed in a saturated clay. Given: depth of embedment of the foundation = 2 m, unit weight of soil = 18 kN/m 3 , and undrained cohesion of clay =...
-
Four point loads with the same magnitude of P are applied as shown in the plan view in Figure P8.1 and are separated by distance b. a. Find the vertical stress increase at a depth of 0.5b below the...
-
A point load of 500 kN is applied at the ground level. Plot the lateral variation of the vertical stress increase at depths of 2 m, 3 m, and 4 m below the ground level.
-
Barnett Brass sells economy door knobs for $15 each. Unit product costs are as follows: Direct materials Direct labor Manufacturing overhead Total $ 3 2 6 $11 An order to purchase 4,000 units was...
-
Culver Inc. presented the following data. Net income $2,610,000 Preferred stock: 51,000 shares outstanding, $100 par, 7% cumulative, not convertible 5,100,000 Common stock: Shares outstanding 1/1...
-
Which of the following comments is true regarding Justin Paperny's comments during his presentation? A. Justin believes that the main reason he was sentenced to prison is because his actions were key...

Study smarter with the SolutionInn App


