Apply the appropriate material balance equations to verify that the lever rule gives the relative amount of
Question:
Apply the appropriate material balance equations to verify that the lever rule gives the relative amount of species in each phase along a tie line as depicted in Figure 8.2.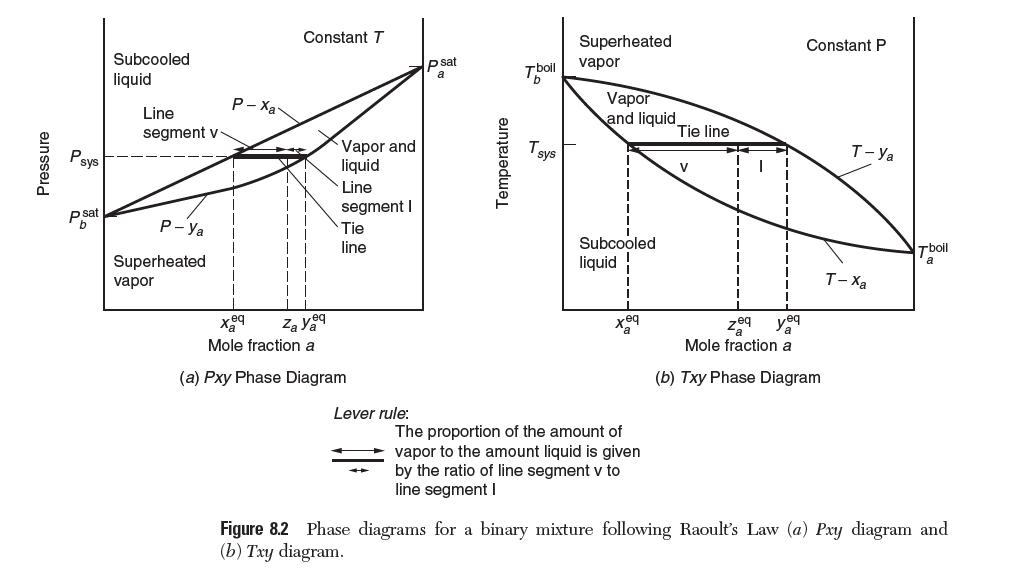
Transcribed Image Text:
Pressure Psys Psat Subcooled liquid Line segment v P-Ya Superheated vapor P-Xa Constant T Za yeq Vapor and liquid Line segment I Tie line Mole fraction a (a) Pxy Phase Diagram Lever rule: psat a Temperature Tboll T sys Superheated vapor Vapor and liquid Subcooled liquid eq The proportion of the amount of vapor to the amount liquid is given by the ratio of line segment v to line segment I Tie line V 1 Constant P Zeq yeq Mole fraction a (b) Txy Phase Diagram T-Ya T-Xa Tboil Figure 8.2 Phase diagrams for a binary mixture following Raoult's Law (a) Pxy diagram and (b) Txy diagram.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
A mass balance o...View the full answer

Answered By

John Aketch
I have a 10 years tutoring experience and I have helped thousands of students to accomplish their educational endeavors globally. What interests me most is when I see my students being succeeding in their classwork. I am confident that I will bring a great change to thins organization if granted the opportunity. Thanks
5.00+
8+ Reviews
18+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Determine each of the following as being either true or false. If it is false, explain the reason why. 2 3-1 0 2 6-2 0 2
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Portray in words what transforms you would have to make to your execution to some degree (a) to accomplish this and remark on the benefits and detriments of this thought.You are approached to compose...
-
Why should one-time write-offs of fixed capital be used in absorption cost systems?
-
What is the reason of the participation rate in Alberta increased to 75 in 2008 then it decreased to 73.8 in 2010?
-
To subscribe to satellite radio, a customer is charged $15 for the activation fee and then an additional $18 per month. Express the total cost C as a function of the number of months of service x. If...
-
A company has made the following payments in respect of electricity and internet expenses: The company has a calendar year end. 1 What balance should have been brought down in the electricity expense...
-
Patients in a hospital are classified as surgical or medical. A record is kept of the number of times patients require nursing service during the night and whether or not these patients are on...
-
7. Epson is considering outsourcing cartridges to a Mexican manufacturer, which has offered to sell an unlimited quantity of cartridges for $5.25 per unit. If Epson accepts the offer, the facilities...
-
A binary vapor mixture contains 48% ethanol (a) in water (b) at 70C. Determine the pressure at which this vapor develops the fi rst drop of liquid. What is the liquid composition? The excess Gibbs...
-
A compressed liquid feed stream containing an equimolar mixture of n-pentane and n-hexane fl ows into a fl ash unit as shown in Figure E8.3 at fl ow rate F. At steady state, 33.3% of the feed stream...
-
Graph each function. Be sure to label key points and show at least two cycles. Use the graph to determine the domain and the range of each function. y = 2 cot x - 1
-
What is classful addressing? Why it is not used anymore? Discuss class B address in brief.
-
6. Factor a* -5a -36.
-
Assume a $70,000 investment and the following cash flows for two alternatives. Year Investment X Investment Y 1 $15,000 $35,000 2 20,000 3 20,000 20,000 20,000 4 20,000 5 15,000 a. Calculate the...
-
A room has dimensions of 9.15 meters by 9.4 meters by 3.68 meters. What is the volume of the room in cm^3?
-
Explain why hosting multiple website by IP address is not preferred method.
-
Explain how each of the following liabilities would be classified in the balance sheet: A note payable of $100,000 due in five years. A note payable of $100,000 payable in annual installments of...
-
Archangel Corporation prepared the following variance report. Instructions Fill in the appropriate amounts or letters for the question marks in the report. ARCHANGEL CORPORATION Variance...
-
Show the conjugate bases of these species: a) H-0-H_bH-0 H CH-N-H d) H-C-C-H H
-
Complete these acid-base equations. Use the curved arrow method to show the electron movement in the reactions. Base Acid a) NH, + H: b) CHO + HO: Conjugate acid Conjugate base
-
Indicate whether each of these species is a Lewis acid, a Lewis base, or both: H 1. a) H-C I H d) CH3-N-H T H b) H-O-H :8-3-6: :ci: e) :CI-AI :CI: H c) H-B 1 H
-
(a) Bonnie contributes land, which she held as inventory (FMV 473,000 and AB 516,000), in exchange for an interest in the JB Partnership. The partnership holds the land as inventory and sells it...
-
Employer health taxes are calculated on: Multiple Choice Calculated on taxable earnings as defined by the jurisdiction T4 employment earnings Earnings up to the annual maximum pensionable earnings...
-
Each tax payment made by most corporations throughout the year a. is a portion of the actual income tax for the previous year. b. is the actual tax on income earned in the previous quarter. c. is a...

Study smarter with the SolutionInn App


