The following data are available for vaporliquid equilibrium of the methane (1) n-pentane (2) binary system at
Question:
The following data are available for vapor–liquid equilibrium of the methane (1) –n-pentane (2) binary system at 37.78°C. Compare how well the van der Waals and Peng–Robinson equations can represent these data using the equation of state method to calculate fugacity coeffi cients of the vapor and the liquid.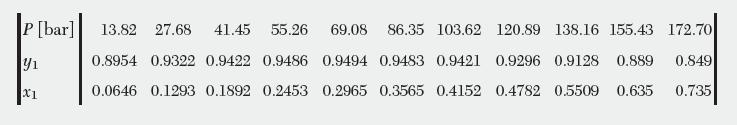
Transcribed Image Text:
P [bar] 13.82 27.68 41.45 55.26 69.08 86.35 103.62 120.89 138.16 155.43 172.70 0.849 0.8954 0.9322 0.9422 0.9486 0.9494 0.9483 0.9421 0.9296 0.9128 0.889 0.0646 0.1293 0.1892 0.2453 0.2965 0.3565 0.4152 0.4782 0.5509 0.635 0.735 y X1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
We can use the solution approach developed in Examples 815 and 816 to calculate the value of pressur...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Repeat Example 8.17 for the PengRobinson equation with values of the binary interaction parameter, k12 of 0.025, 0.05, and 0.10. Example 8.17 The following data are available for vaporliquid...
-
Compute the CLV metric and then apply it in a simple breakeven analysis. The data below show the results of a Starbucks segmentation study. Each column shows the values for a typical customer in each...
-
The directors of Mylo Ltd are currently considering two mutually exclusive investment projects. Both projects are concerned with the purchase of new plant. The following data are available for each...
-
Prepare adjusting journal entries, as needed, considering the account balances excerpted from the unadjusted trial balance and the adjustment data. A. depreciation on fixed assets, $ 8,500 B....
-
Which of the following questions should a marketing researcher ask when trying to establish the reliability of secondary data sources in the international arena? a. How much does the data cost? b....
-
Find for each of the given trigonometric functions. Assume is an acute angle. Round off results. sec = 34.2
-
Your local newsagent has an accounting year ending on 31 December. The following amounts have been paid for gas heating: Required You are required to show the double entries in the light and heat...
-
The partners of Clash Company have decided to liquidate their business. Noncash assets were sold for $125,000. The income ratios of the partners M. Jones, J. Strummer, and P. Simonon are 3:2:3,...
-
Question 20 5 pts When a client prepares summary financial statements: 1. The auditor checks that there are no inconsistencies with the full financial statements. 2. The auditor prepares a...
-
Calculate the equilibrium composition of the two liquid phases in a binary mixture of methyl diethylamine (a) and water (b) at 1 bar and 20C. Solve this problem in two ways; by graphical methods and...
-
Repeat Example 8.15 using the PengRobinson equation of state. Compare your answer to the reported measured value of P = 69.1 bar and y1 = 0.95. Example 8.15 Using the equation of state method,...
-
You have just received notification that you have won the $1 million first prize in the Centennial Lottery. However, the prize will be awarded on your 100th birthday (assuming youre around to...
-
= A membrane process is being designed to recover solute X from a dilute solution where c 3x102kg mol X/m by dialysis through a membrane to a solution where c2 = 0.5 x 102kg mol X/m. The membrane...
-
How do credit rating agencies employ complex analytical models to assess the creditworthiness of corporate entities, and what impact does their evaluation have on the cost of borrowing and capital...
-
What are some factors to consider when choosing a financial advisor? What is the best way to save for retirement, and how much should I be saving each month? How can I improve my credit score, and...
-
what ways do financial institutions utilize advanced risk management techniques such as value-at-risk (VaR), stress testing, and scenario analysis to quantify and mitigate potential credit risk...
-
First, we will perform a quick sanity check using Ohm's Law, i.e. power supply and resistors only, to give ballpark values for Rc and RE in P3. If we are dealing with a 12V power supply and want to...
-
Indicate whether each of the following assets and liabilities should be classified as current or noncurrent: (a) Accounts receivable; (b) Prepaid rent for the next six months; (c) Note receivable due...
-
Rosalie owns 50% of the outstanding stock of Salmon Corporation. In a qualifying stock redemption, Salmon distributes $80,000 to Rosalie in exchange for one-half of her shares, which have a basis of...
-
Show the hybridization at each of the atoms, except H, in these molecules. Indicate the type of each designated bond and the orbital's that are overlapping to form it. : a) H_C_C,H 4 (both) : ...
-
Circle the conjugated pi bonds, if any, in the following compounds? c) CH,CH=CH-C=N b) CH3-CH=CH-C-CH3 d) H-C=C-CH=CH-C-H La e) CH-CH=CH-CH-C-CH3
-
What is the hybridization at the indicated atoms in these compounds? a) CHCH=CH_NHCH3 12 3 c) CH,=CH0CH, 1 2 3 4 5 0: b) CHC0CH, 1 d) 2 1 NH
-
In the dividends-based valuation process discussed in the book and my notes, what does "continuing value" refer to? The value of dividends after the forecast horizon, which in book examples is...
-
The right side of the manufacturing overhead account Blank______. Multiple select question. will usually equal the left side of the account at the end of the year accumulates overhead costs as they...
-
when a practice recognizes and records revenue only when payments or cash are received by the practice and only recognizes and reports an expense when money is actually paid out, they are using what...

Study smarter with the SolutionInn App


