The following solidliquid equilibrium data are available for a binary mixture of C and metastable Fe. From
Question:
The following solid–liquid equilibrium data are available for a binary mixture of C and metastable γ—Fe.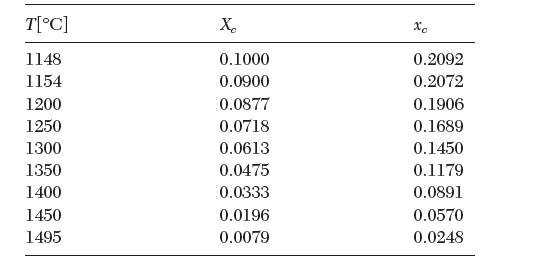
From these data estimate the melting point and enthalpy of fusion of γ—Fe.
Transcribed Image Text:
T[C] 1148 1154 1200 1250 1300 1350 1400 1450 1495 Xc 0.1000 0.0900 0.0877 0.0718 0.0613 0.0475 0.0333 0.0196 0.0079 xc 0.2092 0.2072 0.1906 0.1689 0.1450 0.1179 0.0891 0.0570 0.0248
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
The excess Gibbs energy for a binary mixture of liquid a and liquid b is given by: where T is in [K]. The solids of these species are completely immiscible. The enthalpies of fusion and melting...
-
The following data are available for a binary mixture of ethanol and water at 20C: (a) Make a plot of the partial molar volumes of ethanol and water vs. mole fraction ethanol. (b) What is vmix for an...
-
Assume that the company experienced super-normal growth rate of 30% in the first 3 years and then return to its long-run constant growth rate of 6%. The current dividend is RM2 and the required rate...
-
The monthly earnings of financial analysts are normally distributed with a mean of $5,700. If only 6.68 % of the financial analysts have a monthly income of more than $6,140, what is the value of the...
-
What does it mean to "fix" nitrogen, and what natural processes accomplish nitrogen fixation?
-
Use the function for which the graph is shown in Fig. 3.38 to sketch graphs of the indicated functions. f(x + 2) Fig. 3.38 - 0
-
An asset costs 15,000, has a residual value of 1,000 and an estimated useful life of five years. Using the reducing balance method and a 40 percent rate per annum, what is the assets net book value...
-
Vanessa Noel, owner and manager of Noel Draperies and Window Treatments, has been receiving some complaints from her loyal clientele of interior decorators and home dcor consultants. For example, one...
-
Chance Company had two operating divisions, one manufacturing farm equipment and the other office supplies. Both divisions are considered separate components as defined by generally accepted...
-
When 9 g of urea (CH4N2O) are added to 1 kg of acetone at 1 bar, the boiling point of acetone raises 0.24 K. The normal boiling point of acetone is 329.2 K. From this datum, estimate acetones...
-
A well-insulated container has 1.0 kg ice and 1.0 kg liquid water at 0C in equilibrium. 1.0 kg of liquid ethanol at 0C is added to the system. At equilibrium, what is the fi nal state of the system?...
-
Give the phase for the following states. Discuss.
-
1. Why is steel a good material for cables? 2. What is wrong with a stone suspension bridge? 3. What is reinforced concrete?
-
3 points You are attempting to measure the elastic modulus of a human femur and have attached a strain gauge along the length of the bone. Under a certain load the bone sample expands from 100mm to...
-
An unknown material in the shape of a cylinder with a length of 3.0 m and a diameter of 0.02 m is connected to a constant supply of potential difference equal to 20.0 V. An ammeter measures current...
-
Q1. Q2. Q3. Q4. Q3 Year 3 years ago 2 years last year this year next year Generate forecasts for each of the 4 quarters for the next year by using the following 4 forecasting methods. And compute...
-
A8. In a Si wafer with an area of 1 cm, the recombination time for holes is t=10-6 s and the hole mobility is 480 cmV-s. The Si wafer is uniformly illuminated with UV light of 21016 -2 photons cm s...
-
Selected balance sheet information for the Wolf Company at November 30, and December 31, 2011, is presented below. The company uses the perpetual inventory system and all sales to customers are made...
-
State whether each of the following will increase or decrease the power of a one-way between-subjects ANOVA. (a) The effect size increases. (b) Mean square error decreases. (c) Mean square between...
-
Di-peptides result from the reaction of two amino acids to form an amide. Explain which nitrogen of the following di-peptide is the stronger base: CH,O LI H_NCH, C-NH-CH-C-0
-
Explain which nitrogen in the ring of the amino acid histidine is the stronger base: N- N-H CH + HN CHCO, Histidine
-
The pKa of the carboxylic acid group of acetic acid is 4.7. The pKa of the carboxylic acid group of the conjugate acid of the amino acid alanine is 2.3. Explain the difference in these pKa values....
-
The transactions in the journal and the accounts in the ledger are linked together by a transfer process called:Group of answer choicesaudit trailmigratingpostingbalancing
-
Garbo township recorded it's police departments appropriations for the year ending June 30, 2025. Police appropriations had increased to $7,600,000. During the year's first budgetary month ending...
-
QUALITY INDICATOR: Ninety-five percent of physician appointments/reappointments will be completed within 90 days of receipt of all required materials. medical staff office health information business...

Study smarter with the SolutionInn App


